Research Article - Modern Phytomorphology ( 2026) Volume 20, Issue 1
Therapeutic potential of Moringa oleifera and Curcuma longa on fascioliasis in a murine model
Tarfa A. Altorki1,2, Maimonah Alghanmi1,2, Sarah A. Altwaim3,4, Isra Alsaady1,3, Sara H. Mokhtar1, Bayan Tashkandi1, Hadeel Alsadoun1,5, Rowaida A. Bakri6, Hattan Gattan1,3, Majed H. Wakid1,3*, Rowa Alhabbab1,2 and Ayat Zawawi1,22Vaccines and Immunotherapy Unit, King Fahd Medical Research Center, King Abdulaziz University, Jedda, Saudi Arabia
3Special Infectious Agents Unit, King Fahd Medical Research Center, King Abdulaziz University, Jedda, Saudi Arabia
4Department of Clinical Microbiology and Immunology, Faculty of Medicine, and King Abdulaziz Univers, King Abdulaziz University, Jeddah, Saudi Arabia
5Stem Cell Unit, King Fahd Medical Research Center, King Abdulaziz University, Jeddah, Saudi Arabia
6Department of Parasitology, Faculty of Medicine, Umm Al Qura University, Makkah, Saudi Arabia
Majed H. Wakid, Department of Medical Laboratory Sciences, Faculty of Applied Medical Sciences, King Abdulaziz Unive, P.O. Box 80324, Jeddah 21589, Saudi Arabia, Email: mwakid@kau.edu.sa
Received: 18-Feb-2026, Manuscript No. mp-26-185266; Accepted: 18-Mar-2026, Pre QC No. mp-26-185266 (PQ); Editor assigned: 20-Feb-2026, Pre QC No. mp-26-185266 (PQ); Reviewed: 06-Mar-2026, QC No. mp-26-185266; Revised: 13-Mar-2026, Manuscript No. mp-26-185266 (R); Published: 20-Mar-2026, DOI: 10.5281/zenodo.19081055
Abstract
Fascioliasis is a zoonotic disease that affects millions of people in the Middle East. Therefore, this study aimed to investigate the antiparasitic impact of Moringa oleifera and Curcuma longa on Fasciola hepatica-infected rats. Rats were divided into four groups: Uninfected negative control; infected untreated positive control; infected and treated with C. longa extract; infected and treated with M. oleifera seed extract. The liver enzymatic activity, egg count, and histopathological impact in infected and uninfected groups were determined, and the therapeutic efficacy was assessed. The study outcomes showed that the treated groups had fewer histopathological markers of Fasciola infection than the untreated group. Furthermore, the treated groups had much-reduced amounts of Fasciola egg in feces, and liver enzymes were retrained near the normal range. This effect was more pronounced in the M. oleifera extract group compared to the C. longa extract group. M. oleifera extracts have demonstrated promising efficacy in the treatment of fascioliasis in animal models.
Keywords
Moringa oleifera, Curcuma longa, Fasciola hepatica, Antiparasitic, Fascioliasis
Abbreviations
ALT: Alanine Aminotransferase; AST: Aminotransferase; ALP: Alkaline Phosphatase
Introduction
Foodborne fascioliasis is one of the most prevalent zoonotic parasitic diseases affecting the liver and bile ducts, mainly caused by two Fasciola species: F. hepatica and F. gigantica (Alsulami, et al. 2023, Toulah, et al. 2019, WHO, 2026). Human fascioliasis has recently emerged as a public health problem, with the World Health Organization (WHO) categorizing it among the neglected tropical diseases (WHO, 2025, Mas-Coma, et al. 2022). This infection has caused a global economic burden estimated at more than 90,000 Disability-Adjusted Life Years (DALYs) worldwide (Rosas-Hostos, et al. 2023). Despite strict control measures, recent reports revealed a significant increase in the incidence of Fasciola in Northern Africa, the Middle East, Southeast Asia, and South America (Alsulami, et al. 2022). The rise in cases in Fasciola-endemic regions can be attributed to increased human migration, extreme poverty, deteriorating living conditions, animal exports, and climate change (Ashoor and Wakid, 2023).
The principal etiology of pathological alterations in fascioliasis, specifically the hepatic lesions in infected patients, is the migration, penetration, and prolonged burrowing of juvenile flukes into the liver parenchyma (Alsulami, et al. 2022, Ashoor and Wakid, 2023). These lesions are caused by mechanical damage of the hepatic parenchyma and from destruction caused by molecules secreted by the parasite, which modulate the host’s immune response, resulting in hemorrhage followed by necrosis, fibrosis, and cirrhosis of the liver. Furthermore, this continuous damage can lead to inflammation of the liver and bile ducts, accompanied by hyperplasia of the bile duct epithelium (Toulah, et al. 2019, Alsulami, et al. 2022, Ashoor and Wakid 2023).
Due to the large size of the adult flukes, inflammation and epithelial hyperplasia may occur, which may lead to biliary obstruction, epigastric pain, right upper quadrant abdominal tenderness, biliary colic and jaundice that may lead to hepatosplenomegaly and gallstone formation. Additionally, biochemical changes in infected hosts typically show elevated liver enzyme of serum Alanine Aminotransferase (ALT), Aspartate Aminotransferase (AST) and Alkaline Phosphatase (ALP) (Caravedo and Cabada, 2020, Gandhi, et al. 2019).
Various strategies have been implemented to mitigate the spread of fascioliasis, including improvements in sanitation, chemical fertilizers, food sanitation, health education programs, and triclabendazole chemotherapy (Toulah, et al. 2019, Gandhi, et al. 2019, Merachew and Alemneh, 2020). However, an increase in fascioliasis cases have been observed, indicating rising drug resistance, therefore, researchers are diligently striving to identify alternative therapies to combat the emergence of drug resistance and to develop an effective vaccine (Toulah, et al. 2019, Merachew and Alemneh, 2020).
Medicinal herbs have effectively served as anthelmintics, both as individual remedies and in combination with manufactured medicines (Tagboto and Townson, 2001). Several studies have evaluated the effects of these herbs and plants on various types of flukes, including Fasciola spp. (Toulah, et al. 2019, Mas-Coma et al. 2022, Ullah, et al. 2017). Moringa oleifera is a plant of significant interest to scientists, belongs to the Moringaceae family and is known for its antiepileptic, antiulcer, antipyretic, cholesterol-lowering, diuretic, antidiabetic, and hepatoprotective properties (Alsulami, et al. 2022, Ashoor and Wakid, 2023). M. oleifera has been investigated as an antiparasitic herb against a variety of parasites, including cestodes, trematodes, nematodes and protozoa (Bulus and Addau, 2013, Almanzor, et al. 2014, Abdeltawab, et al. 2025, Konmy, et al. 2023, Budiapsari, et al. 2024, Elghandour, et al. 2023, Nishi, et al. 2021, Rizk, et al. 2023, Saad El-Din, et al. 2023, Hammi, et al. 2020, Xiao, et al. 2020).
The current study aimed to investigate the effects of M. oleifera and C. longa extracts on hepatic histopathological changes and egg shedding in F. hepatica infection. In addition, the activities of pivotal hepatotoxicity biomarkers AST, ALT, and ALP were investigated.
Materials and Methods
Preparation of M. oleifera seed and C. longa methanolic extract
The dried ground seeds of M. oleifera were macerated in 70% methanol (ratio 1:10 w/v) under stirring for 72 hr at room temperature as previously described with simple modification (El-Fakharany, et al. 2024). The filtrate solvent then was evaporated at 40â, and the crude extract was collected and kept at 4°C.
The dried ground rhizomes of C. Longa were macerated in 96% methanol for 72 hr at room temperature as previously described with simple modification (Vanda, et al. 2020).
The filtrate solvent was evaporated at 40â, and the crude extract was collected and kept at 4°C.
Experimental animals design
Twenty-four male albino rats, weighing 180-220 g, were housed under controlled conditions; temperature (22 ± 2°C), 12-hour light-dark cycles, and provided access to food and water as needed.
To ensure that rats were free of any endoparasites, feces were examined microscopically, as previously described, using direct smears, concentration sedimentation method and permanent staining (Garcia, et al. 2017, Bahwaireth and Wakid, 2022, Al-Refai and Wakid, 2024).
The twenty-four rats were randomly allocated into four groups (six in each group); uninfected negative control, infected untreated positive control, infected and treated with C. longa extract (300 mg/kg/day), infected and treated with M. oleifera seed extract (150 mg/kg/day). Treatment with C. longa and M. oleifera began from week 10 to week 12. Treatment doses were determined based on prior in vivo studies that demonstrated antiparasitic and hepatoprotective activities of M. oleifera seed extracts at 100-200 mg/kg and C. longa at 200-300 mg/kg (Mooney, et al. 2009). These doses were chosen as this range showed minimal toxicity and measurable biological effects in rodent models. Treatment extracts with C. longa and M. oleifera were administered orally using oral-gastric gavage to ensure accurate dosing.
Prior to infection, the viability of the metacercaria was confirmed by observing cyst motility and the presence of refractile excretory granules under light microscopy. Only viable/ intact metacercariae were selected. A standardized inoculum of 50 metacercariae per rat was used to ensure consistent infection across all groups.
Parasitological assessment
The number of Eggs Per Gram of stool (EPG) was determined at weeks 9-12 post-infection using the sedimentation technique as described previously by Elmalawany, et al. 2024, with a slight modification. The severity of the infection before treatment was determined by measuring the baseline shedding level at week 9. Weekly monitoring assessed the changes induced by treatment in EPG.
Hepatotoxicity biomarkers
Blood samples for biochemical analysis were drawn from the external jugular vein of the animals at weeks 0, 9 and 12, placed in suitable vacuum tubes, and stored at 4°C. The hepatotoxicity biomarkers AST, ALT, and ALP were measured according to standard procedures using the Dimension® Xpand® Plus Integrated Chemistry System, at weeks 0, 9 and 12.
Histopathological examination
At week 12, the animals were slaughtered, and liver tissue sections were prepared then fixed in 10% formalin. The paraffin-embedded sections were subjected to routine histological examination using Hematoxylin and Eosin (H and E), Gomori’s silver impregnation to detect the degree of fibrosis.
Immunohistochemistry PCNA examination
Liver tissue sections (4 μm) were dewaxed, then rehydrated and treated with 3% hydrogen peroxide to block endogenous peroxidase. Antigen retrieval was performed using microwave heating in citrate buffer (pH 6.0), and the slides were washed with TBS and incubated with rabbit serum to reduce nonspecific binding. The slides were incubated overnight at 4°C with 1:200 mouse monoclonal anti-Proliferating Cell Nuclear Antigen (PCNA), (clone PC10; Dako). Then slides were then washed with TBS and treated with biotinylated rabbit anti-mouse IgG (Dako), followed by 1:300 streptavidin-peroxidase ABC complex (Dako).
DAB chromogen was used to visualize the results, which were then counterstained with hematoxylin, dehydrated and mounted. Positive and negative controls were used. The PCNA labelling index was calculated using the proportion of positive hepatocyte nuclei in five high-power microscopic fields (400x).
Statistical analysis
All statistical analyses were carried out with the Statistical Package for Social Science (SPSS) v. 24. Independent samples t-test, Paired samples t-test, and One-way repeated measurements analysis of variance (ANOVA) were used to assess if there were significant differences between the infected and control groups of animals when P ≤ 0.05, differences were considered significant.
Results
Four groups of albino rats, six in each group, were used to evaluate the effects of M. oleifera and C. longa extracts on F. hepatica infection. Experimentally infected rats with F. hepatica were compared according to the following criteria: Uninfected negative control, infected and untreated positive control, infected and treated with C. longa, and infected and treated with M. oleifera seeds. The treatment of M. oleifera and C. longa extracts was introduced orally from week 10-12 post-infection.
The experiments were carried out by assessing F. hepatica EPG found in rats faces, hepatotoxicity biomarkers AST, ALT, and ALP, from week 9 to week 12. In addition to the histopathological changes at the end of the experiment at week 12 post-infection.
Fasciola EPG
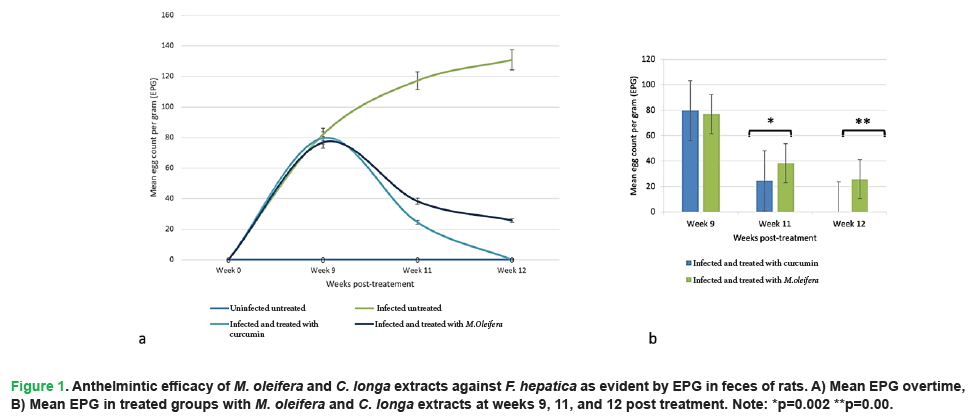
Examination of fecal eggs revealed that all the experimentally infected rats were passing Fasciola eggs by week 9 post infection. Whereas the uninfected negative group did not pass any eggs in their feces. A gradual decrease in Fasciola EPG was detected from week 10 post C. longa and M. oleifera treatments until the end of the experiment (Fig. 1A). Notably, rats treated with M. oleifera showed a significant decrease in EPG in week 11 compared to those treated with C. longa and a complete absence of egg passing was recorded by week 12 post-treatment (Fig. 1B).
Figure 1: Anthelmintic efficacy of M. oleifera and C. longa extracts against F. hepatica as evident by EPG in feces of rats. A) Mean EPG overtime, B) Mean EPG in treated groups with M. oleifera and C. longa extracts at weeks 9, 11, and 12 post treatment. Note: *p=0.002 **p=0.00.
Liver enzymes
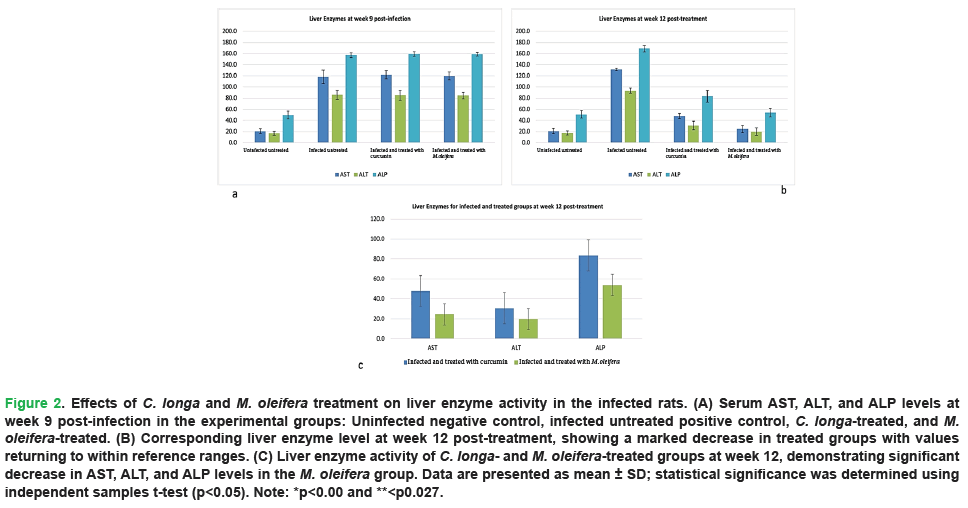
Comparative analysis of the experimental animal groups- uninfected negative control, infected positive control, C. longa-treated, and M. oleifera-treated- demonstrated significant alterations in liver enzymes (AST, ALT, and ALP) activity at weeks 9 post infection, before treatment (Fig. 2A), and at the end of the experiment, week 12 post-treatment (Fig. 2B). At week 9 post-infection, serum liver enzymes (AST, ALT, and ALP) level was significantly elevated in all experimentally infected groups compared to the uninfected control group, indicating hepatic injury associated with F. hepatica infection. In contrast, the uninfected negative control group maintained normal liver enzyme level.
Following treatment, a marked improvement in liver function was observed. By week 12 post-treatment, levels of AST, ALT, and ALP had decreased significantly in both the C. longa- and M. oleifera-treated groups, returning to the normal range. This reduction demonstrates the significant therapeutic effects of both treatments in ameliorating liver damage induced by F. hepatica infection. Furthermore, a direct comparison between the C. longa-treated group and the M. oleifera-treated group at week 12 (Fig. 2C), using an independent samples t-test, revealed a statistically significant difference in liver enzyme activity. Notably, the decline in liver enzyme level was more pronounced in the M. oleifera-treated group, suggesting a stronger hepatoprotective effect compared to C. longa.
Figure 2: Effects of C. longa and M. oleifera treatment on liver enzyme activity in the infected rats. (A) Serum AST, ALT, and ALP levels at week 9 post-infection in the experimental groups: Uninfected negative control, infected untreated positive control, C. longa-treated, and M. oleifera-treated. (B) Corresponding liver enzyme level at week 12 post-treatment, showing a marked decrease in treated groups with values returning to within reference ranges. (C) Liver enzyme activity of C. longa- and M. oleifera-treated groups at week 12, demonstrating significant decrease in AST, ALT, and ALP levels in the M. oleifera group. Data are presented as mean ± SD; statistical significance was determined using independent samples t-test (p<0.05). Note: *p<0.00 and **
Histological results
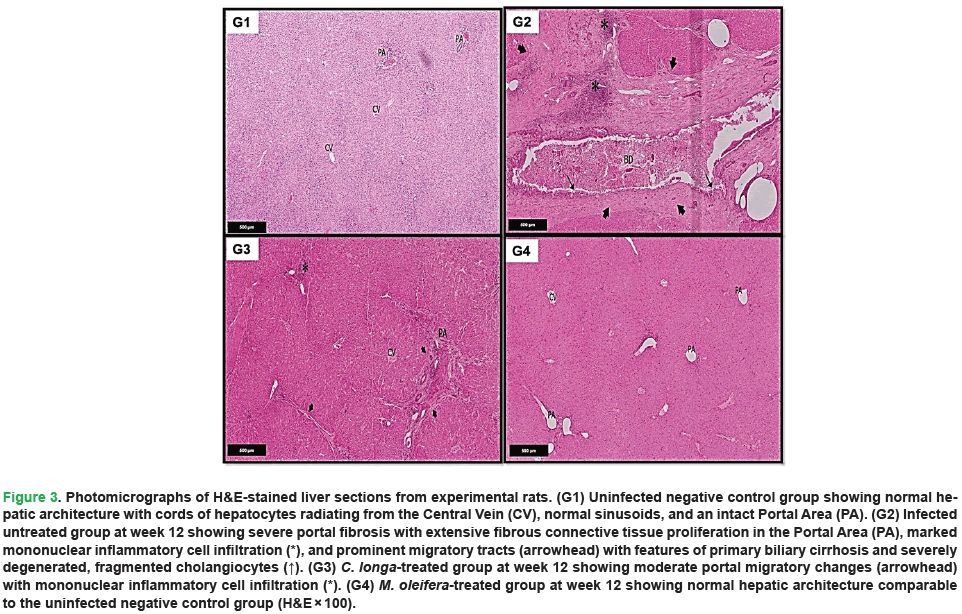
Histopathological analyses confirmed the biochemical findings and revealed distinct differences between the experimental animal groups (Figs. 3-7). Histopathological examination of H&E-stained liver sections from uninfected negative control group demonstrated normal hepatic architecture, characterized by well-organized cords of hepatocytes radiating from the central vein and separated by intact blood sinusoids. The hepatocytes exhibited vesicular nuclei, occasionally with binucleated cells, and normal sinusoidal lining composed of flattened endothelial cells and Kupffer cells. The portal areas showed normal structural components, including branches of the portal vein, hepatic artery, and bile duct, lined by intact columnar cholangiocytes (Fig. 3-5, G1).
Figure 3: Photomicrographs of H&E-stained liver sections from experimental rats. (G1) Uninfected negative control group showing normal hepatic architecture with cords of hepatocytes radiating from the Central Vein (CV), normal sinusoids, and an intact Portal Area (PA). (G2) Infected untreated group at week 12 showing severe portal fibrosis with extensive fibrous connective tissue proliferation in the Portal Area (PA), marked mononuclear inflammatory cell infiltration (*), and prominent migratory tracts (arrowhead) with features of primary biliary cirrhosis and severely degenerated, fragmented cholangiocytes (↑). (G3) C. longa-treated group at week 12 showing moderate portal migratory changes (arrowhead) with mononuclear inflammatory cell infiltration (*). (G4) M. oleifera-treated group at week 12 showing normal hepatic architecture comparable to the uninfected negative control group (H&E × 100).
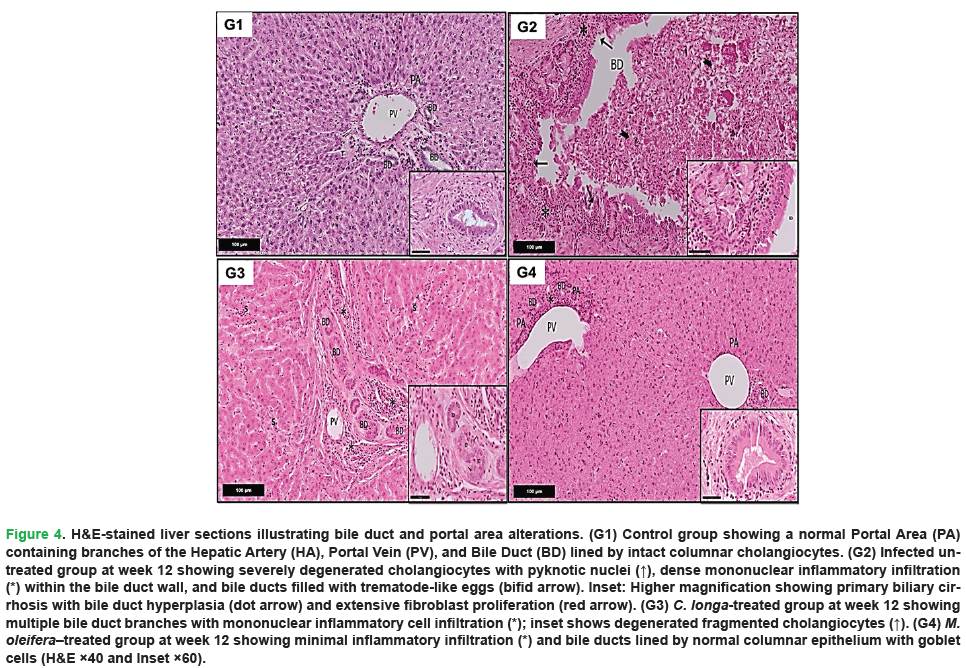
Figure 4: H&E-stained liver sections illustrating bile duct and portal area alterations. (G1) Control group showing a normal Portal Area (PA) containing branches of the Hepatic Artery (HA), Portal Vein (PV), and Bile Duct (BD) lined by intact columnar cholangiocytes. (G2) Infected untreated group at week 12 showing severely degenerated cholangiocytes with pyknotic nuclei (↑), dense mononuclear inflammatory infiltration (*) within the bile duct wall, and bile ducts filled with trematode-like eggs (bifid arrow). Inset: Higher magnification showing primary biliary cirrhosis with bile duct hyperplasia (dot arrow) and extensive fibroblast proliferation (red arrow). (G3) C. longa-treated group at week 12 showing multiple bile duct branches with mononuclear inflammatory cell infiltration (*); inset shows degenerated fragmented cholangiocytes (↑). (G4) M. oleifera–treated group at week 12 showing minimal inflammatory infiltration (*) and bile ducts lined by normal columnar epithelium with goblet cells (H&E ×40 and Inset ×60).
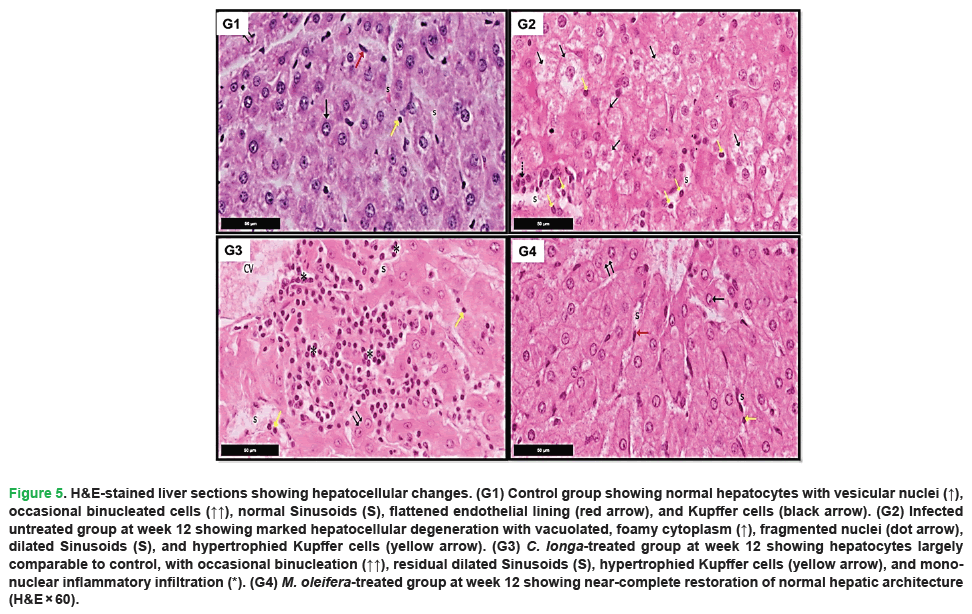
Figure 5: H&E-stained liver sections showing hepatocellular changes. (G1) Control group showing normal hepatocytes with vesicular nuclei (↑), occasional binucleated cells (↑↑), normal Sinusoids (S), flattened endothelial lining (red arrow), and Kupffer cells (black arrow). (G2) Infected untreated group at week 12 showing marked hepatocellular degeneration with vacuolated, foamy cytoplasm (↑), fragmented nuclei (dot arrow), dilated Sinusoids (S), and hypertrophied Kupffer cells (yellow arrow). (G3) C. longa-treated group at week 12 showing hepatocytes largely comparable to control, with occasional binucleation (↑↑), residual dilated Sinusoids (S), hypertrophied Kupffer cells (yellow arrow), and mononuclear inflammatory infiltration (*). (G4) M. oleifera-treated group at week 12 showing near-complete restoration of normal hepatic architecture (H&E × 60).
In contrast, liver sections from infected untreated positive control at 12 weeks showed severe pathological alterations (Figs. 3-5, G2). These included marked portal fibrosis with extensive proliferation of fibrous connective tissue and dense infiltration of mononuclear inflammatory cells. Prominent migratory tracts associated with biliary damage were evident, accompanied by features consistent with primary biliary cirrhosis. Cholangiocytes were severely degenerated and fragmented, with pyknotic nuclei, and bile ducts were frequently dilated and filled with trematode-like eggs. Pronounced bile duct hyperplasia within the portal triads was observed, along with extensive inflammatory infiltration comprising lymphocytes, plasma cells, macrophages, and eosinophils. In addition, the hepatocytes exhibited marked degeneration, characterized by vacuolated foamy cytoplasm and pyknotic nuclei, along with dilated sinusoids containing hypertrophied Kupffer cells.
Liver sections from the C. longa-treated group demonstrated a noticeable improvement in hepatic architecture compared to the untreated infected group (Figs. 3-5, G3). The hepatic cords were generally well organized around sinusoids, which appeared largely normal, although slight sinusoidal dilatation and focal inflammatory cell infiltration persisted in some areas. The portal areas were close to normal but occasionally showed migratory changes, multiple bile duct branches, and moderate mononuclear inflammatory infiltrates. Limited areas of cholangiocyte degeneration were still evident.
Examination of liver sections from the M. oleifera-treated group at week 12 showed substantial restoration of normal hepatic tissues (Figs. 3-5, G4). The hepatocytes were arranged in well-defined, branching cords radiating from the central vein, with minimal inflammatory infiltration in the portal triads. The bile ducts were lined with columnar epithelium containing goblet cells, closely resembling the architecture observed in the uninfected negative control group. Most hepatocytes appeared normal, exhibiting vesicular nuclei with prominent nucleoli and mildly condensed chromatin. The cytoplasm was acidophilic and granular, and normal endothelial cells and Kupffer cells were observed within the sinusoids. The portal triads exhibited normal structural components, including the portal vein, hepatic artery, and bile duct.
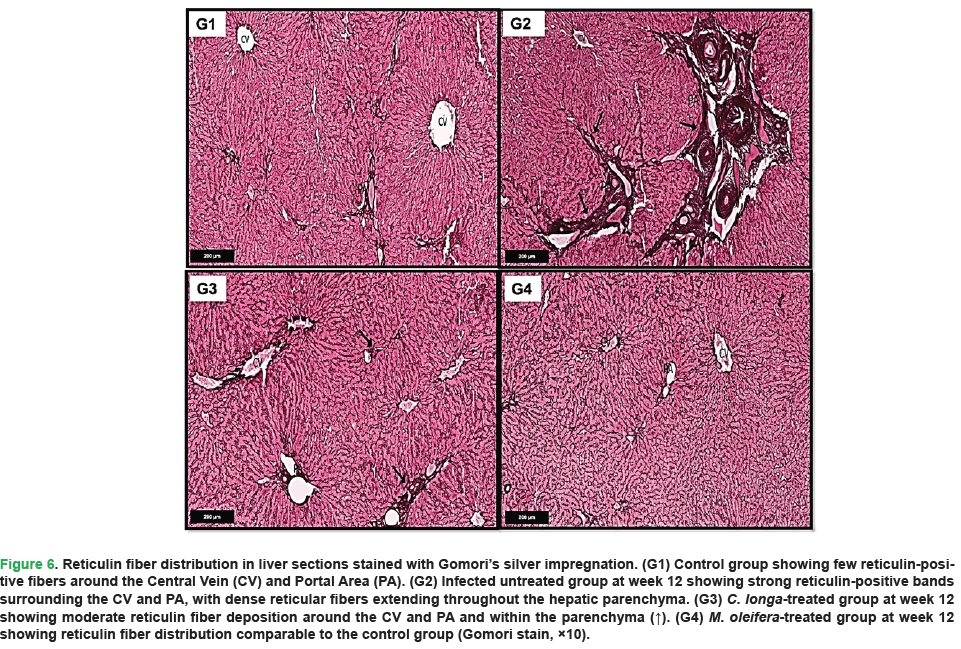
In the uninfected negative control group, liver sections at week 12 stained with Gomori’s silver showed few reticulin-positive fibers surrounding the central veins and portal areas, with minimal reticular framework between hepatocytes (Fig. 6, G1). In contrast, untreated infected rats displayed a marked increase in reticulin-positive bands within the portal areas, with dense reticular fibers extending across the hepatic parenchyma (Fig. 6, G2). C. longa-treated rats exhibited a moderate decrease in reticulin fiber deposition, with moderately expressed reticulin-positive bands around the central veins and portal areas (Fig. 6, G3). Notably, rats treated with M. oleifera exhibited reticulin fiber distribution comparable to the uninfected negative control group, characterized by sparse reticulin-positive bands around the central veins and portal areas and minimal reticular fibers between hepatocytes (Fig. 6, G4).
Figure 6: Reticulin fiber distribution in liver sections stained with Gomori’s silver impregnation. (G1) Control group showing few reticulin-positive fibers around the Central Vein (CV) and Portal Area (PA). (G2) Infected untreated group at week 12 showing strong reticulin-positive bands surrounding the CV and PA, with dense reticular fibers extending throughout the hepatic parenchyma. (G3) C. longa-treated group at week 12 showing moderate reticulin fiber deposition around the CV and PA and within the parenchyma (↑). (G4) M. oleifera-treated group at week 12 showing reticulin fiber distribution comparable to the control group (Gomori stain, ×10).
PCNA immunostaining
Immunohistochemical PCNA staining was performed to assess hepatocellular proliferative activity among the experimental groups. Liver sections from the uninfected negative control group showed an absence of PCNA immunoreactivity, with hepatocytes exhibiting negative nuclear staining (Fig. 7, G1). In contrast, the infected untreated group demonstrated a marked increase in PCNA expression, with numerous hepatocytes exhibiting strong positive nuclear staining distributed throughout the hepatic parenchyma, particularly around the central veins and portal areas (Fig. 7, G2).
In the C. longa-treated group, PCNA expression was noticeably reduced compared to the infected untreated group, with a moderate number of hepatocytes exhibiting positive brown nuclear staining, mainly localized around the central veins and portal areas (Fig. 7, G3). Notably, the M. oleifera-treated group showed the lowest PCNA expression among the treated groups, with only a few hepatocytes displaying weak nuclear immunoreactivity in pericentral and periportal areas (Fig. 7, G4).
Overall, these findings indicate that both C. longa and M. oleifera treatments attenuated the enhanced hepatocellular proliferation induced by F. hepatica infection, with M. oleifera showing a more pronounced effect in restoring hepatocyte proliferative activity toward normal levels.
Figure 7: Immunohistochemical detection of Proliferating Cell Nuclear Antigen (PCNA) in liver sections. (G1) Control group showing negative PCNA staining in hepatocyte nuclei. (G2) Infected untreated group at week 12 showing strong PCNA-positive brown nuclear staining in hepatocytes. (G3) C. longa-treated group at week 12 showing moderate PCNA immunoreactivity. (G4) M. oleifera-treated group at week 12 showing weak PCNA expression, approaching control levels (PCNA immunostaining, ×20).
Description
Our study is the first to investigate the effect of curcumin and moringa extracts on fascioliasis by comparing their anthelmintic effect on hepatic histopathology, egg shedding, and hepatotoxicity biomarkers activity.
Taken together, our histopathological examination revealed that the livers of the untreated infected group suffered from severe histological changes, fibrosis, hypertrophy, and inflammatory infiltration. This may be attributed to the mechanical and toxic effects of the liver flukes, which impacted on the complex vascular and biliary systems that maintain normal liver function (Elmalawany, et al. 2024, Lalor, et al. 2021). On the other hand, the extensive inflammatory infiltration of lymphocytes, eosinophils, and other inflammatory cells in the infected group may be due to histamine release, prompting the body to produce more endogenous histamine along with other inflammatory molecules (Branco, et al. 2018). Our results showed an improvement in liver histopathological alterations after treatment with extracts of C. longa- and M. oleifera, as evidenced by the reduction in fibrosis, hypertrophy, and inflammatory infiltration. This improvement can be attributed to the return to normal liver function following regenerative changes in the liver parenchyma (Elmalawany, et al. 2024, Lalor, et al. 2021).
Previous studies have indicated elevated liver enzyme activity (ALT, AST, and ALP) in individuals infected with Fasciola (Saba, et al. 2004, Saba, et al. 2004). This increase in enzyme levels is attributed to the extensive damage and destruction of liver parenchyma caused by the liver flukes (Kolodziejczyk, et al. 2005, Rubel, et al. 2005). Measurement of hepatotoxicity biomarkers AST, ALT and ALP showed statistically significant differences between the infected group, the group treated with C. longa and the group treated with M. oleifera at week 12 PI. Liver enzyme levels at week 12 were significantly lower in the M. oleifera-treated group than the infected untreated, which also showed a decrease in liver enzyme levels compared to the C. longa-treated group. Thus, M. oleifera treatment significantly reduced liver enzyme level more than treatment with C. longa.
Rehman, et al. 2020 studied the use of curcumin on adult F. gigantica worms and showed that it produced oxidative stress and apoptosis-like effects, disrupting their antioxidant and detoxifying capacities (Rehman, et al. 2020). Another study demonstrated the potential of curcumin and thymoquinone as anthelmintics on F. gigantica adult flukes, affecting their motility and a clear tegument ultrastructure disruptions and erosion of spines in the posterior region and around the acetabulum (Ullah, et al. 2017). Our findings strongly suggest the positive effect of C. longa and M. oleifera seed extract in reducing the pathological outcome of fascioliasis in the treated groups compared to the infected groups. Nevertheless, when comparing the C. longa-treated to M. oleifera-treated groups, the former group showed a more enhanced recovery from infection compared to the latter.
Regarding the number of eggs, there was a statistically significant difference between the group treated with C. longa and that treated with M. oleifera in EPG, with the latter group showing a lower EPG count. Consistent with our findings, a study by Kandil, et al. 2018 reported that rabbits treated with M. oleifera exhibited a significant reduction in the number of eggs in the feces after treatment and throughout the trial, in addition to the eradication of the flukes in the treated group (Kandil, et al. 2018).
Furthermore, in agreement with our observations, El Shanawany, et al. 2019, demonstrated that treatment of naturally infected sheep with M. oleifera enhanced the body weight gain and reduced Fasciola fecal egg count in severely infected groups compared to the untreated group, with a complete reduction in fecal egg count in mildly infected sheep and showed significant decrease in serum IgG, IL-2, and IL-17. Additionally, a study of M. oleifera leaf extracts on F. gigantica non-embryonated and developed eggs in vivo demonstrated a clear concentration-dependent ovicidal effect.
The aqueous extract exhibited the strongest activity, achieving the lowest lethal concentration value (LC50) against not embryonated eggs (Hegazi, et al. 2018).
To overcome the limitations of our study, further research is needed on immunological markers, the mechanism of action of the active ingredients in both extracts, and their antiparasitic efficacy at different concentrations. Additionally, light microscopy, scanning and transmission electron microscopy can be used to obtain more detailed images of the surface morphology and internal structure of liver flukes.
Conclusion
In conclusion, M. oleifera extract has demonstrated a significant anthelmintic effect and aids the host in reversing the Fasciola pathological effect on liver histology and function in rats, making it an outstanding potential for animal therapy. Furthermore, more studies should be undertaken to investigate the active component in M. oleifera to develop a broad-spectrum antiparasitic agent.
Authors Contributions
All authors contributed to the study conception and design. TA, MA, SM and SA designed the study. IA, AZ, RB and BT helped in data curation. SA, AZ, SM and MW carried out formal analysis. MA, MW, RA and HA performed funding acquisition. MA, AZ, SA, IA and HA performed the initial drafting. RB, HA, SA, HG and IA reviewed and edited the initial drafting. MW performed the final critical editing. All authors read and approved the final manuscript.
Ethical Approval
The study was designed with correspondence to the codes of the guidelines for Ethical Conduct in the Care and Use of Animals (ECCUA); experimental conduct and handling were authorized via the Animal Ethics division within the Ethics Committee of Biomedical Research-Faculty of Medicine at King Abdulaziz University, ethical approval number (439/291/616). The experiment was executed in consensus with the guidelines of dealing with experimental animals that are followed in KFMRC, KAU, Jeddah, Saudi Arabia, which are in accordance with the Canadian Council (CC).Acknowledgment
This Project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah, Saudi Arabia under grant no. (G: 214-290-1439). The authors, therefore, acknowledge with thanks DSR for technical and financial support.Funding
This study was funded by the Deanship of Scientific Research (DSR), at King Abdulaziz University, Jeddah, Saudi Arabia, grant number G: 214-290-1439.Conflicts of Interest
The authors declare no conflict of interest.
References
- Abdeltawab MSA, Hamed AMR, Saad El-Din S, Medhat E, Samir M, Mahfoz AM, Mahmoud AWM, Aboulhoda BE, Abdallah HA, Sallam HS, El-Sherbini MS. (2025). Effect of Moringa oleifera and ivermectin nanoparticles on the immunopathological response during experimental trichinosis in mice. Gut Pathog. 17:89.
[Crossref] [Google Scholar] [PubMed]
- Allam G. (2009). Immunomodulatory effects of curcumin treatment on murine schistosomiasis mansoni. Immunobiology. 214:712–27.
[Crossref] [Google Scholar] [PubMed]
- Almanzor IJA, Clemente DE, Fornillos RJC, Gomez MA, Ladiao FRM. (2014). In vivo trials of Moringa oleifera Lam. extracts as antischistosomal treatment on Schistosoma japonicum infected mice. Sanghiran Multidiscip J. 2:49-56.
- Al-Refai MF, Wakid MH. (2024). Prevalence of intestinal parasites and comparison of detection techniques for soil-transmitted helminths among newly arrived expatriate labors in Jeddah, Saudi Arabia. Peer J. 12:e16820.
[Crossref] [Google Scholar] [PubMed]
- Alsulami MN, Mohamed K, Wakid MH, Abdel-Gaber R, Timsah AG, Al-Megrin WAI, Khan A, Elkholy WA, Abdelaal KAA, Elshabrawy HA, El-Kady AM. (2023). Molecular characterization of Fasciola hepatica in sheep based on DNA sequences of ribosomal ITS-1. Infect Drug Resist. 16:6661-6671.
[Crossref] [Google Scholar] [PubMed]
- Alsulami MN, Wakid MH, Al-matary M, bdel-Gaber R, Al-Megrin WAI, Bakhraibah AO, Alanazi AD, Elshabrawy HA, El-Kady AM. (2022). Identification and genetic characterization of Fasciola hepatica isolated from cattle in Jeddah, Saudi Arabia based on sequence analysis of mitochondrial (COI) gene. Infect Drug Resist. 15:4877-4886.
[Crossref] [Google Scholar] [PubMed]
- Ashoor SJ, Wakid MH. (2023). Prevalence and hepatic histopathological findings of fascioliasis in sheep slaughtered in Jeddah, Saudi Arabia. Sci Rep. 13:6609.
[Crossref] [Google Scholar] [PubMed]
- Bahwaireth EO, Wakid MH. (2022). Molecular, microscopic, and immunochromatographic detection of enteroparasitic infections in hemodialysis patients and related risk factors. Foodborne Pathog Dis. 19:830-838.
[Crossref] [Google Scholar] [PubMed]
- Branco ACCC, Yoshikawa FSY, Pietrobon AJ, Sato MN. (2018). Role of histamine in modulating the immune response and inflammation. Mediators Inflamm. 2018:9524075.
[Crossref] [Google Scholar] [PubMed]
- Budiapsari PI, Jaya PK, Dewi PM, Laksemi DA, Horng JT. (2024). Effect of Moringa extract on parasitemia, monocyte activation and organomegaly among Mus musculus infected by Plasmodium berghei ANKA. Narra J. 4:e653.
[Crossref] [Google Scholar] [PubMed]
- Bulus T, Addau FT. (2013). Comparative antitrypanosomal screening of methanolic extracts of Khaya senegalensis and Moringa Oleifera. Sci World J. 8:1-6.
- Caravedo MA, Cabada M. (2020). Human fascioliasis: Current epidemiological status and strategies for diagnosis, treatment, and control. Res Rep Trop Med. 11:149-158.
[Crossref] [Google Scholar] [PubMed]
- Cheraghipour K, Marzban A, Ezatpour B, Khanizadeh S, Koshki J. (2018). Antiparasitic properties of curcumin: A review. AIMS Agric Food. 3:561-578.
- El Shanawany EE, Fouad EA, Keshta HG, Hassan SE, Hegazi AG, Abdel-Rahman EH. (2019). Immunomodulatory effects of Moringa oleifera leaves aqueous extract in sheep naturally co-infected with Fasciola gigantica and Clostridium novyi. J Parasit Dis. 43:583-91.
[Crossref] [Google Scholar] [PubMed]
- El-Fakharany EM, Elsharkawy WB, El-Maradny YA, El-Gendi H. (2024). Moringa oleifera seed methanol extract with consolidated antimicrobial, antioxidant, anti-inflammatory, and anticancer activities. J Food Sci. 89:5130-5149.
[Crossref] [Google Scholar] [PubMed]
- Elghandour MMMY, Maggiolino A, Vázquez-Mendoza P, Alvarado-Ramírez ER, Cedillo-Monroy J, De Palo P, Salem AZM. (2023). Moringa oleifera as a natural alternative for the control of gastrointestinal parasites in equines: A review. Plants. 12:1921.
[Crossref] [Google Scholar] [PubMed]
- Elmalawany AM, Osman GY, Mohamed AH, Khalaf FM, Yassien RI. (2024). Schistosomicidal effects of Moringa oleifera seed oil extract on Schistosoma mansoniâinfected mice. Parasite Immunol. 46:e13070.
[Crossref] [Google Scholar] [PubMed]
- Ezzatkhah F, Mahmoudvand H, Raziani Y. (2023). The role of Curcuma longa essential oil in controlling acute toxoplasmosis by improving the immune system and reducing inflammation and oxidative stress. Front Cell Infect Microbiol. 13:1161133.
[Crossref] [Google Scholar] [PubMed]
- Gandhi P, Schmitt EK, Chen CW, Samantray S, Venishetty VK, Hughes D. (2019). Triclabendazole in the treatment of human fascioliasis: A review. Trans R Soc Trop Med Hyg. 113:797-804.
[Crossref] [Google Scholar] [PubMed]
- Garcia LS, Arrowood M, Kokoskin E, Paltridge GP, Pillai DR, Procop, GW. (2017). Practical guidance for clinical microbiology laboratories: Laboratory diagnosis of parasites from the gastrointestinal tract. Clin Microbiol Rev. 31:e00025-e00017.
[Crossref] [Google Scholar] [PubMed]
- Hammi KM, Essid R, Tabbene O, Elkahoui S, Majdoub H, Ksouri R. (2020). Antileishmanial activity of Moringa oleifera leaf extracts and potential synergy with amphotericin B. S Afr J Bot. 129:67-73.
[Crossref] [Google Scholar] [PubMed]
- Hegazi AG, Abdel Megeed KN, Hassan SE, Abdelaziz MM, Toaleb NI, El Shanawany EE, Aboelsoued D. (2018). Comparative ovicidal activity of Moringa oleifera leaf extracts on Fasciola gigantica eggs. Vet World. 11:215-220.
[Crossref] [Google Scholar] [PubMed]
- HodžiÄ A, Zuko A, AvdiÄ R, AliÄ A, OmeragiÄ J, JažiÄ A. (2013). Influence of Fasciola hepatica on serum biochemical parameters and vascular and biliary system of sheep liver. Iran J Parasitol. 8:92-98.
[Google Scholar] [PubMed]
- Hussein A, Rashed S, El Hayawan I, El-Sayed R, Ali H. (2017). Evaluation of the anti-schistosomal effects of turmeric (Curcuma longa) versus praziquantel in Schistosoma mansoni infected mice. Iran J Parasitol. 12:587-596.
[Google Scholar] [PubMed]
- Kamel HH, Sarhan RM, Saad GA. (2015). Biochemical assessment of oxidative status versus liver enzymes in patients with chronic fascioliasis. J Parasit Dis. 39:628-633.
[Crossref] [Google Scholar] [PubMed]
- Kandil OM, Hassan NMF, Sedky D, Ata EB, Nassar SA, Shalaby HA, Nanev V, Tsocheva-Gaytandzhieva N, Gabrashanska M. (2018). Anthelmintic efficacy of Moringa oleifera seed methanolic extract against Fasciola hepatica. J Parasit Dis. 42:391-401.
[Crossref] [Google Scholar] [PubMed]
- Kolodziejczyk L, Siemieniuk E, Skrzydlewska E. (2005). Antioxidant potential of rat liver in experimental infection with Fasciola hepatica. Parasitol Res. 96:367-372.
[Crossref] [Google Scholar] [PubMed]
- Konmy B, Olounladé PA, Adjobimey T, Dansou CC, Adoho ACC, Tchetan E, Alowanou GG, Azando EVB, Adjanohoun A, Doko Allou SY, Baba-Moussa L. (2023). In vivo anticoccidial activity and immune response of Moringa oleifera and Vernonia amygdalina leaves against Eimeria magna and Eimeria media in rabbits. Front Cell Infect Microbiol. 13:1173138.
[Crossref] [Google Scholar] [PubMed]
- Lalor R, Cwiklinski K, Calvani NED, Dorey A, Hamon S, Corrales JL, Dalton JP, De Marco Verissimo C. (2021). Pathogenicity and virulence of the liver flukes Fasciola hepatica and Fasciola gigantica that cause the zoonosis fasciolosis. Virulence. 12:2839-2867.
[Crossref] [Google Scholar] [PubMed]
- Magalhães LG, Machado CB, Morais ER, Moreira EB, Soares CS, da Silva SH, Da Silva Filho AA, Rodrigues V. (2009). In vitro schistosomicidal activity of curcumin against Schistosoma mansoni adult worms. Parasitol Res. 104:1197-201.
[Crossref] [Google Scholar] [PubMed]
- Mas-Coma S, Valero MA, Bargues MD. (2022). Human and animal fascioliasis: Origins and worldwide evolving scenario. Clin Microbiol Rev. 35:e0008819.
[Crossref] [Google Scholar] [PubMed]
- Merachew W, Alemneh T. (2020). Review on triclabendazole resistance in Fasciola. J Veter Sci Med. 8:1-8.
- Mooney L, Good B, Hanrahan JP, Mulcahy G, de Waal T. (2009). The comparative efficacy of four anthelmintics against a natural acquired Fasciola hepatica infection in hill sheep flock in the west of Ireland. Vet Parasitol. 164:201-205.
[Crossref] [Google Scholar] [PubMed]
- Morais ER, Oliveira KC, Magalhães LG, Moreira EB, Verjovski-Almeida S, Rodrigues V. (2013). Effects of curcumin on the parasite Schistosoma mansoni: A transcriptomic approach. Mol Biochem Parasitol. 187:91-97.
[Crossref] [Google Scholar] [PubMed]
- Nayak A, Gayen P, Saini P, Mukherjee N, Babu SPS. (2012). Molecular evidence of curcumin-induced apoptosis in the filarial worm Setaria cervi. Parasitol Res. 111:1173-1186.
[Crossref] [Google Scholar] [PubMed]
- Nishi L, Sanfelice RADS, da Silva Bortoleti BT, Tomiotto-Pellissier F, Silva TF, Evangelista FF, Lazarin-Bidóia D, Costa IN, Pavanelli WR, Conchon Costa I, Baptista ATA, Bergamasco R, Falavigna-Guilherme AL. (2021). Moringa oleifera extract promotes apoptosis-like death in Toxoplasma gondii tachyzoites in vitro. Parasitol. 148:1447-1457.
[Crossref] [Google Scholar] [PubMed]
- Rehman A, Ullah R, Gupta D, Khan MAH, Rehman L, Beg MA, Khan AU, Abidi SMA. (2020). Generation of oxidative stress and induction of apoptotic like events in curcumin and thymoquinone treated adult Fasciola gigantica worms. Exp Parasitol. 209:107810.
[Crossref] [Google Scholar] [PubMed]
- Rizk MA, El-Sayed SAE, Sayed-Ahmed MZ, Almoshari Y, Alqahtani SS, Ahmad S, Alam N, Marghani BH, Abdelbaset AE, Igarashi I. (2023). Evaluation of the inhibitory effect of Moringa oleifera leaves methanolic extract against in vitro growth of several Babesia species and Theileria equi and the in vivo growth of Babesia microti. J Trop Med. 2023:4285042.
[Crossref] [Google Scholar] [PubMed]
- Rizk MA, El-Sayed SAES, Igarashi I. 2021. Effects of methanolic extract from turmeric (Curcuma longa) against the in vitro multiplication of several Babesia species and Theileria equi. Parasitologia. 1:188-196.
- Rosas-Hostos Infantes LR, Paredes Yataco GA, Ortiz-Martínez Y, Mayer T, Terashima A, Franco-Paredes C, Gonzalez-Diaz E, Rodriguez-Morales AJ, Bonilla-Aldana DK, Barahona LV, Grimshaw AA, Chastain DB, Sillau S, Marcos LA, Henao-Martínez AF. (2023). The global prevalence of human fascioliasis: A systematic review and meta-analysis. Ther Adv Infect Dis. 10:20499361231185413.
[Crossref] [Google Scholar] [PubMed]
- Rubel D, Prepelitchi L, Kleiman F, Carnevale S, Wisnivesky-Colli C. (2005). A focus study from a case of human fascioliasis in Neuquén. Medicina (B Aires). 65:207-212.
[Google Scholar] [PubMed]
- Saad El-Din MI, Gad El-Hak HN, Ghobashy MA, Elrayess RA. (2023). Parasitological and histopathological studies to the effect of aqueous extract of Moringa oleifera Lam. leaves combined with praziquantel therapy in modulating the liver and spleen damage induced by Schistosoma mansoni to male mice. Environ Sci Pollut Res Int. 30:15548-15560.
[Crossref] [Google Scholar] [PubMed]
- Saba R, Korkmaz M, Inan D, MamikoÄlu L, Turhan O, Günseren F, Cevikol C, KabaalioÄlu A. (2004). Human fascioliasis. Clin Microbiol Infect. 10:385-387.
[Crossref] [Google Scholar] [PubMed]
- Sahebi K, Shahsavani F, Mehravar F, Hatam G, Alimi R, Radfar A, Bahreini MS, Pouryousef A, Teimouri A. (2024). In vitro and in vivo anti-parasitic activity of curcumin nano emulsion on Leishmania major (MRHO/IR/75/ER). BMC Complement Med Ther. 24:238.
[Crossref] [Google Scholar] [PubMed]
- Tagboto S, Townson S. (2001). Antiparasitic properties of medicinal plants and other naturally occurring products. Adv Parasitol. 50:199-295.
[Crossref] [Google Scholar] [PubMed]
- Toulah F, Ashoor S, Wakid M, Alshathly M. (2019). In vitro antihelminthic activity of ethanol Zingiber officinale extract on Fasciola gigantica in comparison to triclabendazole. J Egypt Soc Parasitol. 49:599-610.
- Ullah R, Rehman A, Zafeer MF, Rehman L, Khan YA, Khan MA, Khan SN, Khan AU, Abidi SM. (2017). Anthelmintic potential of thymoquinone and curcumin on Fasciola gigantica. PLoS One. 12:e0171267.
[Crossref] [Google Scholar] [PubMed]
- Vanda H, Parindra R, Hambal M, Athaillah F. (2020). Anthelmintic activity of Curcuma Aeruginosa Roxb extract on Fasciola gigantica in vitro. The 1st international conference on veterinary, animal, and environmental sciences (ICVAES 2019). E3S Web of Conferences. 151:01046.
- WHO. (2025). Neglected tropical diseases: Fascioliasis.
- WHO. (2026). Foodborne trematode infections.
- Xiao X, Wang J, Meng C, Liang W, Wang T, Zhou B, Wang Y, Luo X, Gao L, Zhang L. (2020). Moringa oleifera Lam and its therapeutic effects in immune disorders. Front Pharmacol. 11:566783.
[Crossref] [Google Scholar] [PubMed]