Review Article - Modern Phytomorphology ( 2026) Volume 20, Issue 1
Safe usage and therapeutic evaluation of herbal medicines derived from medicinal plants in children: A collaborative review
Maha Ali Aldraimly1*, Nosaiba Basheer Khairi2, Rahaf Humaidan Alamri3, Razan Abdullah Alrehaili4, Somayah Aljuhani5, Shoug Abdulhafith Alahmady2, Lama Ahmed Qarras6, Rahaf Hussain Mufti6 and Yousef Ahmed Sayed Noor Mohammed62Nahdi Medical Company, Madina, Saudi Arabia
3House Health Pharmacy, Madina, Saudi Arabia
4Mouwasat Hospital, Madina,, Saudi Arabia
5Yanbu National Hospital, Yanbu Industrial City, Saudi Arabia
6National Guard Health Affairs, Madina, Saudi Arabia, Saudi Arabia
Maha Ali Aldraimly, Prince Mohammed Bin Abduaziz (PMBAH) Madinah Amunawarah, Saudi Arabia, Email: marsn-2008@htmail.com
Received: 07-Feb-2026, Manuscript No. mp-26-186237; , Pre QC No. mp-26-186237 (PQ); Editor assigned: 10-Feb-2026, Pre QC No. mp-26-186237 (PQ); Reviewed: 24-Feb-2026, QC No. mp-26-186237; Revised: 10-Mar-2026, Manuscript No. mp-26-186237 (R); Published: 17-Mar-2026, DOI: 10.5281/zenodo.19438504
Abstract
Background: Increasing global use of herbal medicines in paediatric populations, concerns regarding safety, dosing, interactions, and regulatory oversight.
Objective: To systematically evaluate evidence on herbal medicine use in children and examine the collaborative role of pediatricians and pharmacists in ensuring safety, efficacy, and rational use.
Methods: Databases searched (PubMed, Scopus, Cochrane, Embase); PRISMA-guided systematic review; inclusion criteria; risk of-bias assessment; qualitative synthesis.
Results: Summarize number of studies included; most commonly used herbs; reported benefits; adverse effects; interaction risks; identified gaps in interprofessional coordination.
Conclusion: Interdisciplinary collaboration improves medication safety, parental counseling, pharmacovigilance, and therapeutic outcomes.
Keywords
Herbal medicines and herbal plants, Interprofessional collaboration, Drug–herb interactions, Pharmacovigilance, Medication safety, Complementary medicine
Introduction
The use of herbal medicines and other Complementary and Alternative Medicine (CAM) therapies in pediatric populations has increased substantially over the past two decades. Globally, parents frequently turn to herbal products for conditions such as upper respiratory tract infections, colic, sleep disturbances, gastrointestinal discomfort, and immune support. According to the World Health Organization (WHO), traditional and herbal medicine remains a primary or complementary healthcare approach for a large proportion of the world’s population, particularly in Asia, Africa, and parts of the Middle East. The WHO Traditional Medicine Strategy 2014–2023 further emphasizes the growing integration of traditional remedies into national health systems and the need to strengthen regulation and safety monitoring (World Health Organization, 2013).
In pediatric care, herbal medicines are often perceived as “natural” and therefore safe. However, children differ significantly from adults in pharmacokinetic and pharmacodynamic profiles. Developmental immaturity of hepatic enzyme systems, renal clearance mechanisms, and blood–brain barrier permeability can alter drug metabolism and increase susceptibility to toxicity (Kearns, et al. 2003). These physiological differences are particularly relevant when herbal products contain bioactive compounds capable of interacting with cytochrome P450 enzymes or other metabolic pathways. For example, St. John’s Wort (Hypericum perforatum) has been shown to induce CYP3A4, potentially reducing the efficacy of concomitant medications (Izzo and Ernst, 2009).
Despite widespread use, robust clinical evidence supporting the efficacy and safety of many herbal products in children remains limited. A systematic review by Posadzki, et al. 2013 highlighted inconsistencies in safety reporting of herbal medicines in pediatric populations, including underreporting of adverse events. Similarly, Smith, et al. 2018 noted that many herbal products lack standardized dosing guidelines, creating challenges for clinicians responsible for ensuring safe administration. Concerns have also been raised regarding contamination, misidentification of plant species, and variability in active ingredient concentration (Ekor, 2014).
The interprofessional collaboration between pediatricians and pharmacists plays a critical role in mitigating these risks. Pediatricians are responsible for clinical diagnosis, treatment planning, and monitoring of therapeutic outcomes. Pharmacists contribute specialized expertise in pharmacokinetics, formulation assessment, herb–drug interaction screening, and medication safety counseling. Emerging literature emphasizes that collaborative healthcare models improve medication reconciliation accuracy, reduce preventable adverse drug events, and enhance parental education (World Health Organization, 2010; Mekonnen, et al. 2016). In the context of herbal medicine, such collaboration is essential because parents may not routinely disclose the use of herbal products unless specifically asked (Robinson and McGrail, 2019).
Given the expanding use of herbal medicines in pediatric care and the documented safety concerns, there is a pressing need to synthesize current evidence on therapeutic outcomes and safety monitoring while clarifying the respective and shared roles of pediatricians and pharmacists. A systematic review examining these collaborative roles can help inform clinical guidelines, strengthen pharmacovigilance systems, and support evidence-based integration of herbal medicine within pediatric healthcare.
Literature Review
This systematic review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta- Analyses (PRISMA 2020) guidelines to ensure transparency, reproducibility, and methodological rigor. A comprehensive literature search was performed across major electronic databases, including PubMed/MEDLINE, Scopus, Embase, Web of Science, and the Cochrane Library. The search covered studies published between January 2010 and February 2026. Keywords and controlled vocabulary (MeSH terms where applicable) included combinations of “pediatric,” “children,” “herbal medicine,” “phytotherapy,” “complementary medicine,” “pharmacist,” “pediatrician,” “drug–herb interaction,” “medication safety,” and “interprofessional collaboration.” Boolean operators were used to refine the search strategy.
Studies were eligible for inclusion if they involved participants aged 0–18 years, evaluated the therapeutic use or safety of herbal medicines, or examined the roles of pediatricians and pharmacists in herbal medication management. Randomized controlled trials, cohort studies, case-control studies, cross-sectional studies, qualitative research exploring professional roles, and systematic reviews were considered. Studies focusing exclusively on adults, non-therapeutic herbal use, opinion pieces without empirical data, and nonpeer- reviewed materials were excluded. Only articles published in English were included.
Titles and abstracts were screened independently by two reviewers, followed by full-text assessment to determine eligibility. Disagreements were resolved through discussion and consensus. Data extraction was performed using a standardized form capturing study characteristics, population details, type of herbal intervention, reported outcomes, adverse effects, and descriptions of healthcare professional involvement. The methodological quality of randomized controlled trials was assessed using the Cochrane risk of bias 2 tool, while observational studies were evaluated using the Newcastle–Ottawa scale. Due to anticipated heterogeneity in study designs and outcome measures, findings were synthesized narratively rather than through meta-analysis.
Overview of common herbal medicines used in children
Herbal medicines are widely used in pediatric populations for both acute and chronic conditions. Parental preference for plantbased therapies is often influenced by cultural traditions, accessibility, and the perception that “natural or herbal” products are safer than synthetic medications. However, clinical evidence, safety monitoring, and dosing standardization vary considerably between herbal products. This section summarizes some of the most commonly used herbal medicines in children and evaluates the current evidence regarding their therapeutic use and safety.
Chamomile (Matricaria chamomilla, of the Asteraceae or Compositae family) is frequently used in infants and young children to manage colic, mild gastrointestinal discomfort, and sleep disturbances. Its therapeutic properties are primarily attributed to flavonoids and apigenin, which exhibit anti-inflammatory and mild sedative effects. Some clinical studies suggest that chamomile preparations, particularly when combined with fennel (Foeniculum vulgare) or lemon balm (Melissa officinalis L.), may reduce infantile colic symptoms (Martinelli, et al. 2017). Adverse events are uncommon but may include allergic reactions, particularly in children with sensitivities to plants in the Asteraceae family (Ekor, 2014). Overall, chamomile appears relatively safe when used short term and in appropriate formulations.
Fennel (Foeniculum vulgare, of the Apiaceae (parsley or carrot) family) is commonly administered for infantile colic and digestive discomfort due to its antispasmodic effects mediated by anethole. Clinical evidence suggests some reduction in colic episodes; however, variability in dosing and formulation limits generalizability (Alexandrovich, et al. 2003). Concerns have been raised about estragole content and its potential carcinogenicity in high doses, although risk appears minimal at therapeutic levels (European Medicines Agency, 2018). Pediatricians and pharmacologists should carefully assess product quality and dosage before recommending use.
Ginger (Zingiber officinale, of the family Zingiberaceae) is used in older children and adolescents primarily for nausea and vomiting, including motion sickness and postoperative nausea. Its antiemetic properties are linked to gingerols and shogaols that influence gastrointestinal motility and serotonin receptors. A growing body of evidence supports ginger’s effectiveness in managing nausea, with favorable safety profiles when used appropriately (Marx, et al. 2013). Gastrointestinal discomfort is the most commonly reported adverse effect. Caution is advised in children taking anticoagulants due to potential antiplatelet activity.
Echinacea (refers to the a genus of herbaceous flowring plants in the family of daisy, Asteraceae) consists of common species prominent for medicine. It includes herbal species such as Echinacea purpurea (purple coneflower), while other such as E. angustifolia and E. pallida. The genus is widely promoted for immune support and prevention of Upper Respiratory Tract Infections (URTIs). Pediatric trials have produced mixed results. Some studies report modest symptom reduction, while others show no statistically significant benefit (Taylor, et al. 2003). Adverse events such as rash and allergic reactions have been documented, particularly in atopic children. Systematic evaluations suggest that while Echinacea is generally well tolerated, evidence for consistent clinical efficacy in children remains limited (Posadzki, et al. 2013).
St. John’s Wort or Hypericum perforatum L., of the family Hypericaceae, is occasionally used in adolescents for mild to moderate depressive symptoms. Although evidence supports its efficacy in adults, pediatric data remain scarce. A significant concern is its interaction potential through induction of cytochrome P450 enzymes, particularly CYP3A4, which can reduce plasma concentrations of various medications including antidepressants and oral contraceptives (Izzo and Ernst, 2009). The risk of serotonin syndrome when combined with Selective Serotonin Reuptake Inhibitors (SSRIs) further underscores the need for medical supervision (Tab. 1).
| Herbal medicine | Common indications | Evidence level* | Reported risks | Interaction concerns |
| Chamomile | Colic, mild insomnia | Moderate | Allergic reactions | Minimal |
| Fennel | Infantile colic | Limited–Moderate | Possible estragole exposure | Minimal |
| Ginger | Nausea, vomiting | Moderate | GI discomfort | Anticoagulant interaction |
| Echinacea | URTIs | Limited | Rash, allergy | Low |
| St. John’s Wort | Adolescent depression | Limited (pediatric) | Serotonin syndrome risk | High (CYP3A4 induction) |
| Note: *Evidence level based on availability of pediatric clinical trials and systematic reviews. | ||||
Table 1. Common herbal medicines used in pediatric practice.
Overall, the safety profiles of commonly used herbal medicines or medicinal plants in children appear favorable when products are standardized and used appropriately. Nevertheless, variability in formulation, risk of contamination, and limited high-quality pediatric randomized controlled trials remain persistent challenges (Ekor, 2014). Underreporting of adverse events and lack of disclosure to healthcare providers may further complicate clinical oversight. These concerns highlight the importance of structured involvement of pediatricians, pharmacologists, and pharmacists in evaluating herbal use, screening for interactions, and counseling caregivers.
Safety and pharmacological considerations
Safety evaluation of herbal medicines derived from medicinal plants in children requires careful attention to pediatric pharmacology, product quality, dosing uncertainty, and the risk of herb–drug interactions. Unlike conventional pediatric medicines, many herbal products are marketed as dietary supplements or traditional remedies and may not undergo the same pre-marketing testing for efficacy, safety, or standardized dosing. This creates a complex clinical situation in which exposure can be unpredictable, while children especially infants may be physiologically more vulnerable to adverse effects.
From a pharmacokinetic perspective, pediatric patients differ from adults in absorption, distribution, metabolism, and excretion. Gastric pH, intestinal motility, and gut microbiota composition change with age and can influence the bioavailability of herbal constituents. Body water and fat proportions shift across childhood, affecting distribution volumes of hydrophilic or lipophilic compounds. Most importantly, hepatic metabolic capacity and renal clearance are developmentally regulated, meaning that the same herbal dose may produce higher systemic exposure in younger children compared with older adolescents. These developmental changes complicate extrapolation of adult dosing to pediatric populations and strengthen the rationale for conservative dosing and close monitoring when herbs are used therapeutically (Kearns, et al. 2003).
Herb-drug interactions represent one of the most clinically significant safety issues. Interactions may occur through pharmacokinetic mechanisms (altered metabolism or transport) or pharmacodynamic mechanisms (additive, synergistic, or antagonistic effects). A well-documented example is St. John’s Wort (Hypericum perforatum), which can induce CYP3A4 and P-glycoprotein, reducing plasma concentrations of multiple drugs and potentially compromising treatment efficacy. Although St. John’s Wort is more commonly used in adults, adolescent use has been reported, and the interaction risk is clinically relevant for antidepressants, antiepileptics, immunosuppressants, and hormonal contraceptives (Izzo and Ernst, 2009). Similarly, ginger may increase bleeding tendency in susceptible patients through antiplatelet effects, which becomes important when children are receiving anticoagulants or have bleeding disorders (Marx, et al. 2013). These examples illustrate why pharmacists’ interaction screening and pediatricians’ clinical oversight are essential whenever herbal products are used alongside prescription medicines.
Adverse reactions to herbal medicines in children may be underrecognized because caregivers often do not report herbal use unless asked directly, and clinicians may not routinely document it. A key challenge is that many adverse effects are nonspecific such as rash, gastrointestinal upset, irritability, or sleep disturbance making causality assessment difficult. Systematic evidence indicates that adverse event reporting for herbal medicine is inconsistent, and underreporting remains a major barrier to accurate pediatric risk estimation (Posadzki, et al. 2013). In addition, children with asthma, atopy, or pollen allergies may be more likely to develop hypersensitivity reactions to certain botanicals (for example, chamomile in Asteraceae-sensitized individuals), reinforcing the need for allergy history screening (Ekor, 2014).
Quality control is another central safety concern. The chemical composition of herbal products can vary by plant species, geographic origin, harvesting conditions, and extraction methods. Even products sold under the same common name may differ in active constituent concentration. Beyond variability, contamination and adulteration can pose direct harms. Reports in the broader herbal safety literature describe contamination with heavy metals, pesticide residues, and microbial pathogens, and occasional adulteration with pharmaceutical agents to enhance perceived efficacy. Such risks are particularly relevant for children because lower body weight increases dose-per-kilogram exposure, potentially magnifying toxicity. Regulatory reviews and pharmacovigilance discussions emphasize that improving manufacturing standards, authentication of plant materials, and post-marketing surveillance is critical for safer pediatric use (Ekor, 2014).
Dosing and formulation issues add further complexity. Many herbal products lack pediatric-specific dosing guidance supported by trials. Liquid preparations (teas, syrups, tinctures) vary widely in concentration, and alcohol-based tinctures may be inappropriate for younger children. Essential oils raise special concerns: They can be highly concentrated and have been associated with poisoning when ingested unintentionally, and topical exposure may cause dermatitis or systemic effects in infants. For these reasons, safety-focused practice generally favors standardized, child-appropriate formulations with clear labeling and avoidance of highly concentrated oils for internal use in children unless supported by robust clinical guidance. Finally, safe pediatric use depends on structured clinical processes. Medication reconciliation should explicitly include herbs and supplements; clinicians should ask caregivers about product name, dose, frequency, brand, and purpose. Pharmacists can assist by evaluating product quality, verifying dosing logic, screening interaction risks, and educating caregivers on warning signs that require medical attention. Pediatricians are critical for assessing whether herbal use is appropriate for the child’s condition, ruling out serious illness, and monitoring outcomes. At the systems level, strengthening herb-related adverse event reporting and integrating herb–drug interaction resources into clinical workflows can reduce preventable harm and support evidence-based, collaborative care (World Health Organization, 2010).
Role of pediatricians
Pediatricians play a central role in ensuring the safe and evidence-based use of herbal medicines in children. As primary clinicians responsible for diagnosis, therapeutic decision-making, and longitudinal follow-up, pediatricians are uniquely positioned to evaluate whether herbal therapies are appropriate, potentially beneficial, or contraindicated in specific clinical contexts. Given the growing prevalence of Complementary and Alternative Medicine (CAM) use in pediatric populations, routine inquiry about herbal product use should be integrated into comprehensive medical history-taking (Robinson and McGrail, 2019).
One of the pediatrician’s key responsibilities is to assess the clinical indication for herbal use and to distinguish between self- limited conditions and potentially serious illnesses requiring conventional treatment. For example, symptoms such as persistent fever, prolonged cough, or weight loss may reflect underlying pathology that should not be managed solely with herbal preparations. Clinical oversight ensures that herbal use does not delay diagnosis or evidence-based therapy. The American Academy of Pediatrics (AAP) has emphasized the importance of open dialogue between clinicians and families regarding complementary medicine to prevent unsafe practices and promote informed decision-making (American Academy of Pediatrics, 2017).
Another critical role of pediatricians involves evaluating potential safety risks based on the child’s age, comorbidities, and 4 current medications. Children with chronic illnesses such as epilepsy, asthma, autoimmune disorders, or cardiac disease may be particularly vulnerable to herb–drug interactions or disease exacerbation. Pediatricians must review concurrent medications and consult interaction data where appropriate. As documented in the literature, failure to disclose herbal use can increase the likelihood of preventable adverse events, particularly in children receiving prescription therapies (Izzo and Ernst, 2009). Encouraging parents to disclose all products, including over-the-counter supplements and traditional remedies, is therefore an essential communication task.
Monitoring and follow-up also fall within the pediatrician’s professional responsibilities. If a herbal product is used, documentation should include the product name, dosage, duration, and source. Pediatricians should evaluate therapeutic response objectively and assess for adverse reactions. Given that adverse events associated with herbal medicines are often underreported, systematic documentation contributes to improved pharmacovigilance and patient safety (Posadzki, et al. 2013). Where adverse reactions are suspected, appropriate reporting to national pharmacovigilance systems is recommended.
Education and counseling represent another important dimension of the pediatrician’s role. Many caregivers perceive herbal products as inherently safe, yet variability in product quality and lack of standardized dosing may increase risk. Pediatricians can provide balanced, evidence-informed guidance, explaining both potential benefits and uncertainties. Such communication should respect cultural beliefs while emphasizing safety, scientific evidence, and the child’s best interests. The World Health Organization has highlighted the need for integrating traditional medicine into healthcare systems through professional oversight, ensuring that use is guided by trained practitioners rather than informal advice alone (World Health Organization, 2013).
Finally, pediatricians are key advocates for interdisciplinary collaboration. Working closely with pharmacists enhances medication reconciliation, interaction screening, and formulation assessment. Through collaborative care models, pediatricians and pharmacists can reduce preventable medication-related harm and improve the quality of pediatric healthcare delivery (World Health Organization, 2010). In this context, pediatricians serve not only as clinical decision-makers but also as coordinators of safe, child- centered care when herbal medicines are incorporated into treatment plans.
Role of Pharmacists (or Herbal pharmacists)
Pharmacists play a critical role in promoting the safe and rational use of herbal medicines in pediatric populations. As medication experts, pharmacists possess specialized knowledge in pharmacokinetics, pharmacodynamics, formulation science, and drug–herb interactions, positioning them as essential partners in pediatric care. Given the increasing use of Complementary and Alternative Medicine (CAM) among children, pharmacists are often the most accessible healthcare professionals to whom parents turn for advice regarding over-the-counter herbal products. Their involvement can significantly reduce preventable medication-related harm and improve therapeutic outcomes.
One of the primary responsibilities of pharmacists is comprehensive medication reconciliation. This process includes actively inquiring about herbal supplements, traditional remedies, and over-the-counter products, as caregivers may not routinely disclose them to physicians (Robinson and McGrail, 2019). Pharmacists are trained to identify potential pharmacokinetic and pharmacodynamic interactions between herbal and conventional medicines. For instance, St. John’s Wort may induce cytochrome P450 enzymes, reducing the plasma concentration of several medications, while herbs such as ginkgo or ginger may increase bleeding risk when combined with anticoagulants (Izzo and Ernst, 2009). In pediatric patients receiving chronic therapies such as antiepileptics, immunosuppressants, or psychotropic medications interaction screening is especially critical.
Another essential role of pharmacists involves evaluating product quality and formulation suitability. Unlike conventional pharmaceuticals, many herbal products vary in active ingredient concentration, purity, and manufacturing standards. Variability in extraction methods and lack of standardization can lead to inconsistent dosing and unpredictable effects. Literature has documented concerns regarding contamination with heavy metals, pesticide residues, and undeclared pharmaceutical ingredients in certain herbal preparations (Ekor, 2014). Pharmacists can assess product labeling, verify reputable manufacturers, and advise families to avoid non- standardized or unverified products, thereby minimizing safety risks.
Pediatric dosing considerations further highlight the pharmacist’s expertise. Many herbal remedies lack pediatric-specific dosing guidelines, and extrapolating adult doses to children can lead to underdosing or toxicity. Pharmacists can assist in adjusting doses based on weight and age where evidence exists and recommend safer alternatives when insufficient data are available. They are also positioned to advise on appropriate dosage forms, such as avoiding alcohol-based tinctures in young children and discouraging ingestion of concentrated essential oils due to toxicity concerns.
Pharmacists also contribute significantly to caregiver education. Counseling should include clear information about potential side effects, warning signs requiring medical attention, proper administration techniques, storage conditions, and expected duration of use. Importantly, pharmacists can emphasize that “natural” does not automatically mean safe and encourage communication between caregivers and pediatricians. The World Health Organization has underscored the importance of collaborative, interprofessional approaches in improving medication safety and healthcare quality (World Health Organization, 2010).
Finally, pharmacists play a vital role in pharmacovigilance. Reporting suspected adverse reactions associated with herbal medicines strengthens national safety monitoring systems and contributes to evidence generation. Given the documented underreporting of herbal-related adverse events (Posadzki, et al. 2013), pharmacist engagement in surveillance systems enhances patient safety at both individual and public health levels. Through systematic interaction screening, quality assessment, caregiver education, and collaboration with pediatricians, pharmacists are integral to ensuring that herbal medicine use in children aligns with evidence-based and safety-focused care standards.
Interprofessional collaboration model
The safe integration of herbal medicines into pediatric care requires more than isolated clinical decision-making; it depends on structured interprofessional collaboration between pediatricians and pharmacists. As herbal use among children increases globally, coordinated models of care become essential to ensure safety, optimize therapeutic outcomes, and prevent avoidable adverse events. The World Health Organization (WHO) emphasizes that interprofessional collaborative practice improves healthcare quality, strengthens patient safety, and enhances system efficiency (World Health Organization, 2010). In the context of pediatric herbal medicine, this collaborative approach becomes particularly important due to developmental pharmacological variability and limited pediatric-specific evidence.
Interprofessional collaboration begins with transparent communication and systematic medication reconciliation. Parents frequently use herbal remedies without informing healthcare providers, either because they perceive them as harmless or because they are not directly asked (Robinson and McGrail, 2019). A collaborative model requires that pediatricians routinely inquire about herbal use during clinical assessments, while pharmacists reinforce this inquiry when dispensing medications or providing counseling. Establishing a shared culture of disclosure reduces hidden exposure and allows for proactive interaction screening.
The second core component involves shared clinical decision-making. Pediatricians assess the clinical indication, disease severity, and appropriateness of herbal therapy in relation to conventional treatment. Pharmacists complement this by evaluating pharmacokinetic considerations, checking herb drug interactions, reviewing dosing appropriateness, and verifying product quality. Evidence shows that pharmacist participation in medication safety initiatives significantly reduces medication discrepancies and adverse drug events during care transitions (Mekonnen, et al. 2016). Extending this collaborative practice to herbal products strengthens pediatric medication safety frameworks.
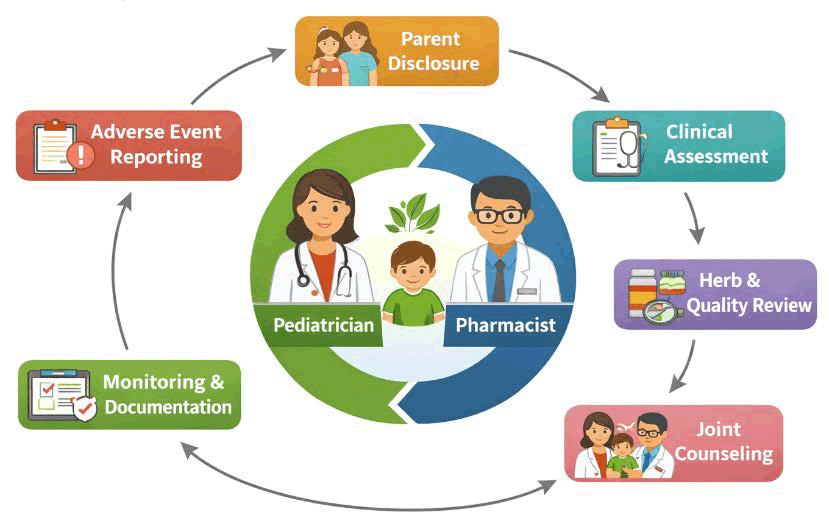
Monitoring and follow-up represent another critical pillar of collaboration. Once herbal therapy is initiated or disclosed, both professionals share responsibility for tracking therapeutic response and adverse reactions. Pediatricians evaluate clinical progress and ensure that herbal use does not delay essential medical interventions. Pharmacists can monitor adherence, identify emerging side effects, and report suspected adverse reactions to pharmacovigilance systems. Given documented gaps in herbal adverse event reporting (Posadzki, et al. 2013), coordinated reporting processes significantly contribute to public health surveillance and safety evidence. Education and caregiver counseling further illustrate the synergy between pediatricians and pharmacists. Pediatricians provide condition-specific guidance, explaining when herbal use may be supportive and when it is not recommended. Pharmacists reinforce this education with detailed counseling about administration techniques, potential side effects, interaction risks, and product storage. This dual reinforcement improves caregiver understanding and reduces misinformation, especially in communities where herbal medicine is culturally embedded. The WHO Traditional Medicine Strategy highlights the importance of integrating traditional therapies within regulated, professional healthcare systems to enhance safety and accountability (World Health Organization, 2013). Institutional integration also strengthens collaborative models. Electronic Health Records (EHRs) that include structured fields for documenting herbal products facilitate shared access to patient information. Embedding herb–drug interaction databases into clinical decision-support systems further enhances safety. Regular interdisciplinary meetings and clinical protocols for herbal use can clarify roles, reduce ambiguity, and support standardized care pathways (Fig. 1).
Figure 1. Interprofessional pediatric herbal care model.
Ultimately, the interprofessional collaboration model transforms herbal medicine management from informal advice-based use into structured, evidence-guided clinical practice. By combining diagnostic expertise, pharmacological knowledge, safety monitoring, and caregiver education, pediatricians and pharmacists create a comprehensive framework that prioritizes the child’s wellbeing. Such collaboration not only reduces risk but also fosters trust, transparency, and informed decision-making in pediatric healthcare.
Regulatory and ethical considerations
The regulation of herbal medicines used in children varies significantly across jurisdictions, creating challenges for consistent safety standards and clinical oversight. In many countries, herbal products are classified as dietary supplements rather than pharmaceutical drugs, meaning they are not required to undergo the same pre-marketing evaluation for efficacy, safety, and quality. This regulatory distinction has direct implications for pediatric use, where developmental vulnerability increases the potential impact of dosing inconsistencies or contamination. The World Health Organization (WHO) has recognized these concerns and emphasized the need for national policies that strengthen quality control, safety monitoring, and appropriate integration of traditional medicine into healthcare systems (World Health Organization, 2013).
In the United States, herbal products are regulated under the Dietary Supplement Health and Education Act (DSHEA), which places the primary responsibility for safety on manufacturers rather than requiring pre-approval by the Food and Drug Administration (FDA). In contrast, the European Medicines Agency (EMA) has established the Committee on Herbal Medicinal Products (HMPC), which evaluates traditional herbal medicinal products and issues scientific monographs to guide safe use, including pediatric considerations where data are available. Despite these efforts, pediatric-specific clinical trials for many herbal preparations remain limited, leaving regulatory bodies reliant on traditional use evidence rather than robust randomized pediatric data (European Medicines Agency, 2018).
From an ethical perspective, the principle of non-maleficence — “do no harm” — is central to decisions about herbal medicine use in children. Because children cannot provide fully informed consent, healthcare providers must act in the child’s best interest while ensuring that parents receive clear, evidence-based information. When efficacy data are weak or inconsistent, clinicians face the ethical challenge of balancing respect for parental preferences with professional responsibility to recommend safe and effective therapies. Transparent communication about uncertainties, potential risks, and interaction concerns supports ethically sound shared decision-making (American Academy of Pediatrics, 2017).
The principle of beneficence further requires clinicians to evaluate whether herbal therapy offers meaningful benefit beyond placebo or supportive care. If a product lacks sufficient evidence but poses minimal risk, cautious short-term use under monitoring may be ethically acceptable in certain circumstances. However, herbal use that delays diagnosis or replaces evidence-based treatment for serious conditions violates ethical standards of pediatric care. Pediatricians and pharmacists therefore share responsibility for ensuring that herbal products complement rather than substitute necessary medical interventions.
Justice and equity considerations are also relevant. In many cultures, herbal medicine is deeply embedded in traditional health practices. Disregarding these practices may undermine trust between families and healthcare providers. Conversely, failure to regulate products effectively may disproportionately expose vulnerable populations to low-quality or contaminated preparations. The WHO’s framework for integrating traditional medicine advocates for culturally sensitive but scientifically grounded regulation to promote equitable healthcare access and patient safety (World Health Organization, 2010).
Finally, pharmacovigilance represents both a regulatory and ethical obligation. Underreporting of herbal-related adverse events weakens evidence generation and delays identification of safety signals (Posadzki, et al. 2013). Healthcare professionals have an ethical duty to report suspected adverse reactions and contribute to post-marketing surveillance systems. Strengthening reporting frameworks and encouraging interdisciplinary documentation can enhance public health protection.
In summary, regulatory variability, limited pediatric-specific evidence, and ethical complexities necessitate careful oversight of herbal medicine use in children. Coordinated action among policymakers, pediatricians, and pharmacists is essential to align cultural practices with evidence-based standards, protect vulnerable pediatric populations, and uphold ethical principles in clinical care.
Discussion
This systematic review examined the safety, therapeutic evaluation, regulatory considerations, and interprofessional roles associated with the use of herbal medicines in pediatric populations. The findings indicate that while several herbal products such as chamomile, ginger, fennel, and Echinacea are commonly used in children and generally demonstrate favorable short-term safety profiles, the strength and consistency of pediatric-specific evidence remain limited. Variability in formulation, dosing uncertainty, and inconsistent adverse event reporting represent persistent safety challenges. These findings are consistent with broader literature highlighting methodological weaknesses in pediatric herbal trials and the underreporting of safety outcomes (Posadzki, et al. 2013; Ekor, 2014).
A key theme emerging from this review is the gap between widespread community use of herbal remedies and the limited clinical integration of such use into structured pediatric care. Many parents perceive herbal medicines as safe because they are “natural,” yet developmental pharmacology demonstrates that children’s metabolic pathways differ significantly from adults, potentially altering risk profiles (Kearns, et al. 2003). Without professional oversight, herbal use may result in unintended herb–drug interactions or delay appropriate treatment for serious conditions. Therefore, the need for collaborative safety monitoring is particularly strong in pediatrics compared with adult populations.
The review also underscores the critical importance of systematic medication reconciliation and structured inquiry. Studies indicate that caregivers often do not disclose herbal use unless directly asked (Robinson and McGrail, 2019). This communication gap can undermine safe prescribing practices and obscure potential causes of adverse reactions. Incorporating routine screening questions about herbal products into pediatric assessments, along with pharmacist-led verification during dispensing and counseling, can reduce hidden exposure and strengthen safety culture. Interprofessional collaboration emerges as a central solution to these challenges. Evidence from medication safety research shows that pharmacist involvement in care transitions and medication review significantly reduces discrepancies and preventable adverse drug events (Mekonnen, et al. 2016). Applying this collaborative model specifically to herbal medicine management provides an evidence-informed pathway to enhance pediatric safety. Pediatricians contribute diagnostic judgment and clinical monitoring, while pharmacists add pharmacological expertise, product quality evaluation, and interaction screening. Together, they can offer balanced, culturally sensitive counseling that neither dismisses parental beliefs nor compromises scientific standards.
Regulatory variability further complicates pediatric herbal medicine use. In many jurisdictions, herbal products are regulated differently from conventional pharmaceuticals, limiting pre-marketing evidence requirements. While the World Health Organization advocates integration of traditional medicine within regulated healthcare systems, implementation remains inconsistent across countries (World Health Organization, 2013). Strengthening regulatory oversight and pharmacovigilance systems is essential, particularly for pediatric populations where safety margins may be narrower. Another important issue is the lack of high-quality randomized controlled trials focused specifically on children. Many existing studies extrapolate adult data or rely on traditional-use evidence rather than pediatric efficacy trials. Future research should prioritize age-stratified dosing studies, standardized formulations, and long-term safety evaluations. Multicenter collaboration could help overcome sample size limitations and improve generalizability. Additionally, integrating herb–drug interaction databases into electronic health records may enhance clinical decision-making and prevent adverse outcomes.
This review has several limitations. Heterogeneity in study designs, outcome measures, and herbal preparations limited the possibility of quantitative synthesis. The inclusion of English-language publications may have excluded relevant data from regions where herbal medicine use is particularly prevalent. Nevertheless, the narrative synthesis provides an integrative perspective that combines clinical pharmacology, regulatory analysis, and interprofessional collaboration literature. While herbal medicines remain widely used in pediatric care, ensuring safety and therapeutic appropriateness requires structured interprofessional engagement. Collaborative models that integrate pediatricians and pharmacists into assessment, counseling, monitoring, and reporting processes offer the most promising pathway to align traditional practices with evidence-based, child-centered healthcare. Strengthening research, regulation, and interdisciplinary communication will be essential to safeguarding pediatric populations in the evolving landscape of complementary medicine.
Conclusion
The increasing use of herbal medicines in pediatric populations reflects cultural practices, parental preferences, and the perception that natural products are inherently safe. However, this review demonstrates that although several herbal remedies commonly used in children appear to have relatively favorable short-term safety profiles, substantial gaps remain in pediatric-specific clinical evidence, dosing standardization, quality control, and pharmacovigilance reporting. Variability in product composition, potential herb–drug interactions, and underreporting of adverse effects present ongoing safety concerns that require structured professional oversight.
Children represent a pharmacologically vulnerable population due to developmental differences in metabolism, organ function, and drug disposition. Consequently, extrapolating adult data to pediatric patients is scientifically insufficient and ethically problematic. The findings of this review highlight that safe integration of herbal medicine into pediatric care cannot rely solely on traditional use or caregiver perception. Instead, it requires systematic clinical assessment, evidence-based evaluation, and interdisciplinary collaboration.
The collaborative model between pediatricians and pharmacists provides a practical and evidence-informed framework to enhance safety, support therapeutic evaluation, and promote transparent communication with caregivers. Pediatricians contribute diagnostic evaluation, clinical monitoring, and treatment prioritization, while pharmacists provide expertise in interaction screening, dosing verification, product quality assessment, and medication counseling. Together, they create a structured, child-centered approach that aligns cultural practices with scientific standards and ethical principles Strengthening regulatory oversight, improving documentation systems, and promoting research specifically designed for pediatric populations are essential steps in minimizing preventable harm. Ultimately, integrating herbal medicine within coordinated healthcare systems rather than managing it informally offers the most effective path toward safeguarding pediatric health while respecting parental preferences.
Recommendations
Based on the findings of this systematic review, several key recommendations emerge:
• Routine herbal use screening: Pediatric assessments should systematically include direct questions about herbal and dietary supplement use to reduce nondisclosure and hidden interaction risks.
• Strengthening interprofessional collaboration: Healthcare institutions should implement structured collaboration protocols between pediatricians and pharmacists, including shared documentation systems and clinical consultation pathways for herbal product evaluation.
• Integration of herb–drug interaction databases: Electronic health record systems should incorporate validated herb–drug interaction tools to support real-time safety decision-making.
• Standardization and quality assurance: Policymakers and regulatory bodies should enhance manufacturing oversight, product labeling standards, and pediatric-specific safety guidance for herbal medicines.
• Enhanced pharmacovigilance reporting: Healthcare professionals should actively report suspected herbal-related adverse events to national surveillance systems to strengthen safety data.
• Pediatric-specific clinical research: Future studies should prioritize randomized controlled trials with age-stratified dosing, long-term safety monitoring, and standardized formulations.
• Caregiver education programs: Structured educational initiatives led jointly by pediatricians and pharmacists should address misconceptions about “natural safety” and promote informed decision-making.
• Ethically guided shared decision-making: Clinical discussions regarding herbal use should balance cultural sensitivity with scientific transparency, ensuring that decisions prioritize the best interest of the child.
References
- Acampora A, Ruini L, Mattia G, Pratesi CA, Lucchetti MC. (2022). Towards carbon neutrality in the agri-food sector: Drivers and barriers. Resour Conserv Recycl. 189:106755.
- Alexandrovich I, Rakovitskaya O, Kolmo E, Sidorova T, Shushunov S. (2003). The effect of fennel (Foeniculum vulgare) seed oil emulsion in infantile colic: A randomized, placebo-controlled study. Altern Ther Health Med. 9:58–61.
[Google Scholar] [PubMed]
- American Academy of Pediatrics. (2017). Complementary and integrative medicine in pediatrics. Pediatrics. 140:e20171958.
- Ekor M. (2014). The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol. 4:177.
[Crossref] [Google Scholar] [PubMed]
- European Medicines Agency. (2018). Assessment report on Foeniculum vulgare Miller, fructus. Committee on Herbal Medicinal Products (HMPC), EMA.
- European Medicines Agency. (2018). Assessment report on herbal medicinal products. Committee on Herbal Medicinal Products (HMPC), EMA.
- Izzo AA, Ernst E. (2009). Interactions between herbal medicines and prescribed drugs. Drugs. 69:1777–1798.
[Crossref] [Google Scholar] [PubMed]
- Kearns GL, Abdel-Rahman SM, Alander SW, Blowey DL, Leeder JS, Kauffman RE. (2003). Developmental pharmacology—Drug disposition, action, and therapy in infants and children. N Engl J Med. 349:1157–1167.
[Crossref] [Google Scholar] [PubMed]
- Marx W, McCarthy AL, Ried K, McKavanagh D, Vitetta L, Sali A, Isenring E, Lohning A. (2013). The effect of ginger on nausea and vomiting in adults and children: A systematic review. Nutr Rev. 71:245–254.
- Martinelli M, Ummarino D, Giugliano FP, Sciorio E, Tortora C, Bruzzese D, Berni Canani R, Staiano A. (2017). Efficacy of a standardized herbal extract in infantile colic. J Pediatr. 186:137–142.
- Mekonnen AB, McLachlan AJ, Brien JA. (2016). Pharmacy-led medication reconciliation programmes at hospital transitions: A systematic review and meta-analysis. J Clin Pharm Ther. 41:128–144.
[Crossref] [Google Scholar] [PubMed]
- Posadzki P, Watson L, Ernst E. (2013). Adverse effects of herbal medicines: An overview of systematic reviews. Clin Med. 13:7–12.
[Crossref] [Google Scholar] [PubMed]
- Robinson A, McGrail MR. (2019). Disclosure of CAM use to medical practitioners: A review of qualitative and quantitative studies. Complement Ther Med. 42:134–142.
[Crossref] [Google Scholar] [PubMed]
- Taylor JA, Weber W, Standish L, Quinn H, Goesling J, McGann M, Calabrese C. (2003). Efficacy and safety of echinacea in treating upper respiratory tract infections in children. JAMA. 290:2824–2830.
[Crossref] [Google Scholar] [PubMed]
- World Health Organization. (2010). Framework for action on interprofessional education and collaborative practice.
- World Health Organization. (2013). WHO traditional medicine strategy 2014–2023.