Research Article - Modern Phytomorphology ( 2023) Volume 17, Issue 1
Phytochemical analysis of Unani herbal formulation "Niswani tea"
Amira Saba*, Syeda sameena aziz, Afshan Jabeen and Najam RehanAmira Saba, Department of Chemistry, Anwarul Uloom College, Mallepally, Hyderabad, T.S-500001, India, Email: amirasaba8@gmail.com
Received: 23-Jan-2023, Manuscript No. mp-23-88996; Accepted: 05-Feb-2023, Pre QC No. mp-23-88996(PQ); Editor assigned: 26-Jan-2023, Pre QC No. mp-23-88996(PQ); Reviewed: 29-Jan-2023, QC No. mp-23-88996(Q); Revised: 03-Feb-2023, Manuscript No. mp-23-88996(R); Published: 15-Feb-2023, DOI: 10.5281/zenodo.7939670
Abstract
Phytochemicals are bioactive compounds obtained from the plants and are widely applied in the traditional unani herbal medicine. Herbal medicine drugs have been used widely in many countries because of its availability, less cost effectiveness and safer than the synthetic drug. These herbal medicines are used by the local people to cure the various diseases which include the major diseases such as Diabetes Mellitus, Cancer, HIV etc. knowledge of phytochemicals constituents present in the unani herbal drugs will support in treating the disease with better therapeutic efficacy. Realizing this, and effort has been made for the screening of various phytochemicals such as coumarins, anthocyanins, leucoanthocyanins, fatty acids, steroids, saponins, terpenoids, quinones, tannins, phlobatannins, phenolic compounds, flavonoids, alkaloids and determination of ash content are present in Niswani tea, unani herbal medicine. It was concluded that the plants studied were rich in phytochemicals with significant pharmacological and medicinal applications.
https://www.mobafire.com/profile/fethiyeboatcharter12-1124680?profilepage https://www.youmagine.com/fethiyeboatcharter12/designs https://www.myminifactory.com/users/Fethiyeboatcharter12 http://qooh.me/Fethiyeboat https://linktr.ee/fethiyeboatcharter12 https://pubhtml5.com/homepage/uqugb/ https://telegra.ph/Fethiyeboatcharter12-10-07 https://www.diggerslist.com/65211afd8d020/about https://allmyfaves.com/Fethiyeboatcharter12 https://www.metal-archives.com/users/Fethiyeboatcharter12 https://www.catchafire.org/profiles/2518744/ https://www.fimfiction.net/user/643834/Fethiyeboatcharter12 https://www.hebergementweb.org/members/fethiyeboatcharter12.562911/ https://www.sqlservercentral.com/forums/user/fethiyeboatcharter12 https://www.twitch.tv/fethiyeboatcharter12/about https://www.roleplaygateway.com/member/Fethiyeboatcharter12/ https://www.provenexpert.com/fethiyeboatcharter12/?mode=preview https://www.intensedebate.com/people/Fethiyeboat https://www.indiegogo.com/individuals/35456265 https://visual.ly/users/fethiyeboatcharter/portfolio https://slides.com/fethiyeboatcharter12 https://letterboxd.com/fethiyeboatchar/ https://fliphtml5.com/dashboard/public-profile/ambnf https://community.windy.com/user/fethiyeboat https://speakerdeck.com/fethiyeboatcharter12 https://trello.com/u/fethiyeboatcharter https://3dwarehouse.sketchup.com/user/a29ef5cb-e718-45a6-a3a3-dc77f6114304/Fethiyeboatcharter12-F https://www.wattpad.com/user/Fethiyeboatcharter12 https://www.goodreads.com/user/show/170514584-fethiyeboatcharter12 https://gravatar.com/fethiyeboatcharter https://www.mapleprimes.com/users/Fethiyeboatcharter12 https://medium.com/@fethiyeboatcharter https://www.ted.com/profiles/45238235 https://www.infragistics.com/community/members/8fb19b684b50b847b2b604e31b8858534f8d18b5?_ga=2.37173475.2130373168.1696671819-632808545.1696671819 https://www.metooo.io/u/652128ef119b751a01cdf6a6 https://app.roll20.net/users/12506970/fethiyeboatcharter12-f https://list.ly/fethiyeboatcharter/activity https://giphy.com/channel/Fethiyeboatcharter12 https://www.tumblr.com/fethiyeboatcharter12 https://www.creativelive.com/student/fethiyeboatcharter12?via=accounts-freeform_2 https://gab.com/Fethiyeboatcharter12 https://www.flickr.com/people/199294447@N04/ https://seedandspark.com/user/fethiyeboatcharter12-01hc4tshz91eyd5mhkekg9mvr7 https://os.mbed.com/users/fethiyeboatcharter12/ https://calis.delfi.lv/blogs/lietotajs/318049-fethiyeboatcharter12/ https://notionpress.com/author/933846 https://my.desktopnexus.com/Fethiyeboatcharter12/ https://guides.co/a/fethiyeboatcharter12-fethiyeboatcharter12/ https://www.bahamaslocal.com/userprofile/1/250276/Fethiyeboatcharter12.html http://molbiol.ru/forums/index.php?showuser=1302412 https://www.credly.com/users/fethiyeboatcharter12-fethiyeboatcharter12/badges https://ko-fi.com/fethiyeboatcharter12#paypalModal https://devpost.com/fethiyeboatcharter?ref_content=user-portfolio&ref_feature=portfolio&ref_medium=global-nav https://www.redbubble.com/people/Fethiyeboat/shop?asc=u https://hub.docker.com/u/fethiyeboatcharter12 http://onlineboxing.net/jforum/user/editDone/254330.page https://micro.blog/Fethiyeboatcharter12 http://www.effecthub.com/user/3729585 http://hawkee.com/profile/5142290/ https://wefunder.com/fethiyeboatcharter12 https://www.facer.io/user/oYTJXrdB85 https://mastodon.online/@Fethiyeboatcharter12 https://sketchfab.com/Fethiyeboatcharter12 https://profiles.wordpress.org/fethiyeboatcharter12/ https://dzone.com/users/5004503/fethiyeboatcharter12.html https://www.mobafire.com/profile/dentalcenterinistanbul12-1126843?profilepage https://www.youmagine.com/dentalcenterinistanbul12/designs https://www.myminifactory.com/users/Dentalcenterinistanbul12 http://qooh.me/Dentalcent https://pubhtml5.com/homepage/awjam/ https://telegra.ph/Dentalcenterinistanbul12-10-18 https://www.diggerslist.com/652fce0d4e58d/about https://allmyfaves.com/Dentalcenterinistanbul12 https://www.catchafire.org/profiles/2530237/ https://www.fimfiction.net/user/647505/Dentalcenterinistanbul12 https://www.hebergementweb.org/members/dentalcenterinistanbul12.567651/ https://www.sqlservercentral.com/forums/user/dentalcenterinistanbul12 https://www.twitch.tv/dentalcenterinistanbul12/about https://www.roleplaygateway.com/member/Dentalcenterinistanbul12/ https://www.provenexpert.com/dentalcenterinistanbul12/?mode=preview https://www.intensedebate.com/people/dentalcenterr https://www.indiegogo.com/individuals/35565363 https://visual.ly/users/dentalcenterinistanbul/portfolio https://slides.com/dentalcenterinistanbul12 https://letterboxd.com/dentalcenterini/ https://fliphtml5.com/dashboard/public-profile/uezzd https://community.windy.com/user/dentalcenterin https://speakerdeck.com/dentalcenterinistanbul12 https://3dwarehouse.sketchup.com/user/cf451399-53bc-4d31-9faf-0a3d991b657f/Dentalcenterinistanbul12-D https://www.wattpad.com/user/Dentalcenterinistanb https://www.goodreads.com/user/show/170837175-dentalcenterinistanbul12 https://gravatar.com/dentalcenterinistanbul https://www.mapleprimes.com/users/Dentalcenterinistanbul12 https://medium.com/@dentalcenterinistanbul https://www.ted.com/profiles/45329577 https://www.infragistics.com/community/members/6888d7ee81952716bf633b6d4e35a805ccc6f664 https://www.metooo.io/u/652fdd8e79c47c11a2f64290 https://app.roll20.net/users/12549184/dentalcenterinistanbul12-d https://list.ly/dentalcenterinistanbul/activity https://giphy.com/channel/Dentalcenterinistanbul12 https://www.tumblr.com/blog/dentalcenterinistanbul12 https://www.creativelive.com/student/dentalcenterinistanbul12?via=accounts-freeform_2 https://gab.com/Dentalcenterinistanbul12 https://www.flickr.com/people/199352431@N02/ https://seedandspark.com/user/dentalcenterinistanbul12-01hd1gdgzksen3y4xz31cmbanc https://os.mbed.com/users/dentalcenterinistanb/ https://calis.delfi.lv/blogs/lietotajs/320045-dentalcenterinistanbul12/ https://notionpress.com/author/938143 https://my.desktopnexus.com/Dentalcenterinistanbul12/ https://guides.co/a/dentalcenterinistanbul12-dentalcenterinistanbul12/ https://www.bahamaslocal.com/userprofile/1/254247/Dentalcenterinistanbul12.html http://molbiol.ru/forums/index.php?showuser=1304433 https://www.credly.com/users/dentalcenterinistanbul12-dentalcenterinistanbul12/badges https://devpost.com/dentalcenterinistanbul?ref_content=user-portfolio&ref_feature=portfolio&ref_medium=global-nav https://hub.docker.com/u/dentalcenterinistanbul12 http://onlineboxing.net/jforum/user/editDone/256030.page https://micro.blog/Dentalcenter http://www.effecthub.com/user/3730799 http://hawkee.com/profile/5210865/ https://wefunder.com/dentalcenterinistanbul12 https://www.facer.io/user/LtJSQQxXiv https://mastodon.online/@Dentalcenterinistanbul12 https://sketchfab.com/Dentalcenterinistanbul12 https://profiles.wordpress.org/dentalcenterr/ https://dzone.com/users/5009302/dentalcenterinistanbul12.html https://www.metal-archives.com/users/Dentalcenterinistanbul12 https://ko-fi.com/dentalcenterinistanbul12#paypalModal https://www.redbubble.com/people/Dentalcenterin/shop?asc=u https://trello.com/u/dentalcenterinistanbul https://linktr.ee/marisdentist12 https://www.mobafire.com/profile/dentalclinicmarmaris12-1124946?profilepage https://www.youmagine.com/dentalclinicmarmaris12/designs https://www.myminifactory.com/users/Dentalclinicmarmaris12 http://qooh.me/Dentalcliniccc https://linktr.ee/dentalclinicmarmaris12 https://pubhtml5.com/homepage/vguev/ https://telegra.ph/Dentalclinicmarmaris12-10-09 https://www.diggerslist.com/6523b5fb88341/about https://allmyfaves.com/Dentalclinicmarmaris12 https://www.catchafire.org/profiles/2520311/ https://www.fimfiction.net/user/644350/Dentalclinicmarmaris12 https://www.hebergementweb.org/members/dentalclinicmarmaris12.563625/ https://www.sqlservercentral.com/forums/user/dentalclinicmarmaris12 https://www.twitch.tv/dentalclinicmarmaris12/about https://www.roleplaygateway.com/member/Dentalclinicmarmaris12/ https://www.provenexpert.com/dentalclinicmarmaris12/?mode=preview https://www.intensedebate.com/people/dentalcliniccc https://www.indiegogo.com/individuals/35472275 https://visual.ly/users/dentalclinicmarmaris/portfolio https://slides.com/dentalclinicmarmaris12 https://letterboxd.com/dentalclinicmar/ https://fliphtml5.com/dashboard/public-profile/zyofe https://community.windy.com/user/dentalcliniccc https://speakerdeck.com/dentalclinicmarmaris12 https://trello.com/u/dentalclinicmarmaris https://3dwarehouse.sketchup.com/user/956c9e20-3db8-466f-bc70-c903b5c5baf4/Dentalclinicmarmaris12-D https://www.wattpad.com/user/Dentalclinicmar https://www.goodreads.com/user/show/170567262-dentalclinicmarmaris12 https://gravatar.com/dentalclinicmarmaris https://www.mapleprimes.com/users/Dentalclinicmarmaris12 https://medium.com/@dentalclinicmarmaris https://www.ted.com/profiles/45253014 https://www.infragistics.com/community/members/ce27f374981a18f23d521975a7367213d41589b9?_ga=2.234072734.1427803247.1696842611-150604057.1696842611 https://www.metooo.io/u/6523c82f79c47c11a2f0ad92 https://app.roll20.net/users/12515324/dentalclinicmarmaris12-d https://list.ly/dentalclinicmarmaris/activity https://giphy.com/channel/Dentalclinicmarmaris12 https://www.tumblr.com/dentalclinicmarmaris12 https://www.creativelive.com/student/dentalclinicmarmaris12?via=accounts-freeform_2 https://gab.com/Dentalclinicmarmaris12 https://www.flickr.com/people/199318845@N06/ https://seedandspark.com/user/dentalclinicmarmaris12-01hc9wxrxeh68sdwmapdstysd3 https://os.mbed.com/users/dentalclinicmarmaris/ https://calis.delfi.lv/blogs/lietotajs/318363-dentalclinicmarmaris12/ https://notionpress.com/author/934484 https://my.desktopnexus.com/Dentalclinicmarmaris12/ https://guides.co/a/dentalclinicmarmaris12-dentalclinicmarmaris12/ https://www.bahamaslocal.com/userprofile/1/250830/Dentalclinicmarmaris.html http://molbiol.ru/forums/index.php?showuser=1302701 https://www.credly.com/users/dentalclinicmarmaris12-dentalclinicmarmaris12/badges https://ko-fi.com/dentalclinicmarmaris12#paypalModal https://devpost.com/dentalclinicmarmaris?ref_content=user-portfolio&ref_feature=portfolio&ref_medium=global-nav https://www.redbubble.com/people/Dentalclinicmar/shop?asc=u https://hub.docker.com/u/dentalclinicmarmaris12 http://onlineboxing.net/jforum/user/editDone/254618.page https://micro.blog/Dentalclinicmar http://www.effecthub.com/user/3729764 http://hawkee.com/profile/5153741/ https://wefunder.com/dentalclinicmarmaris12 https://www.facer.io/user/3NxPOYhyW2 https://mastodon.online/@Dentalclinicmarmaris12 https://sketchfab.com/Dentalclinicmarmaris12 https://profiles.wordpress.org/dentalclinicmarmaris12/ https://dzone.com/users/5004687/dentalclinicmarmaris12.html https://www.metal-archives.com/users/Dentalclinicmarmaris12
Keywords
Phytochemicals, unani herbal medicine, drug, therapeutic, blood pressure, heart care, capsules, flavonoid, saponins, tannins
Introduction
Phytochemicals are bioactive molecules that are also referred to as secondary metabolites that are derived from plants. Primary metabolites and Secondary metabolites are the two types of metabolites generated in plants (Bansode T.S. et al., 2015). Primary metabolites are required for a plant's normal metabolism including growth and development. They may be unnecessary for secondary metabolites generated by plants. These are produced in nearly every part of the plant including the bark, leaves, stem, root, flower, fruits and seeds. Phytochemicals have been utilized as traditional herbal medicine for some years now all over the world. As a result, both the pharmaceutical industry and researchers place a higher focus on phytochemical research. These phytochemicals which are found in many plant sections are also employed by indigenous peoples to treat various ailments (Ugochukwu S.C. et al., 2013). These are also frequently utilized in the agricultural sector. Drugs, flavouring agents, perfumes, color, pigments, insecticides and food additives all rely on secondary metabolites for their synthesis. Many medicines generated from secondary metabolites are simply synthetic alterations or duplicates of these naturally occurring compounds (Hussain M.S et al., 2012).
Following are the commercial importance of some phytochemicals
1. Pesticides such as nicotine, pyrethrins, and rotenone are used in small amounts (Balandrin M.F et al., 1985).
2. Tannins are commonly used as an astringent (Ashok P.K et al., 2012).
3. Quinones, such as hypericin, are antibacterial (Tiwari P et al., 2011).
4. Secondary metabolites are used to investigate diverse metabolic processes using pharmacological techniques. Diterpene esters for example, formed from the latices of certain Euphorbia species are potent irritants and co-carcinogens, making them important in chemical carcinogenesis investigations (Balandrin M.F et al., 1985; Hecker E et al. 1977).
Secondary metabolites have aroused a lot of interest in their research production and they're being investigated extensively as a source of medicinal compounds (Gracelin D.H et al., 2013). As a result, evaluating phytochemicals from various different Unani Herbal Medicine is qualitatively screened for phytochemicals from various different Unani Herbal Medicine extracts appeared to be important. In the present study, Unani Herbal medicine is qualitatively screened for phytochemicals using standard chemical tests.
Secondary metabolites such as saponins, tannins and flavonoids have been discovered to have antidiabetic, anti-inflammatory, hepatoprotective, anti-hyperlipidemia, diuretic, and antibacterial properties (Modi D.C et al., 2010). The Unani Herbal Medicine Niswani Tea used in the production of medicines for a number of infectious diseases including chronic ulcers, leucorrhoea, pyorrhea and fungal infections of the skin. It is good for girl’s/women care formulated with ashoka and kalongi Berg-e-Heena and Gull-e-Nilofer, it is very effective caffeine free herbal remedies for Heart Care and Blood pressure. The barks, seeds and flowers of the tree are helpful in preparing capsules and tonics to solve various gynecological problems of women. It was found that flavonoid is present abundantly in all species. The objective of the present study was to screen such a phytochemicals analysis (Gul R et al., 2017).
Materials and Methods
Drug material
The required Niswani Tea Unani Herbal Medicine is a product of Halal Herbal Remedies Hyderabad 500005 Telangana, India.
Preparation of Unani herbal medicine extract (Niswani Tea)
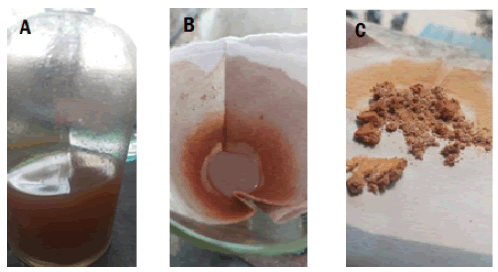
The herbal medicine extract of Niswani tea is prepared using the hot water extraction technique. The powder extract of Niswani Tea was kept in an appropriately labelled plastic bottle. 5gm of powder extract was weighed using an electronic weighing balance, dissolved in a 25 ml of sterile water and then boiled at 50°C-60°C for 30 minutes on water bath. The extract was filtered through Whatman No.1 filter paper and centrifuged the filtrate at 2500 rpm for 15 minutes. For further investigation of phytochemical analyses, the extract was kept in sterile bottle at 4°C-8°C in refrigerator (Savithramma N et al., 2011)(Fig. 1(A-E).

Figure 1: (A)Medicine extract (B) Concentration of extract (C) Filtration (D) Centrifugation (E) Collection of filtrate.
Phytochemical analysis
Preliminary qualitative screening for phytochemicals, of Unani herbal medicine Niswani Tea was carried out with the following test methods.
Test for Coumarins
2 ml of extract was treated with 3 ml of 10% NaOH. The formation of yellow colour indicating the presence of coumarins. The presence of lactone is a key structural motif of coumarins which are hydrolysed once attacked by a strong nucleophile like NaoH into water-soluble salt of cis-cinnamic acid derivatives. Acidification of these salt results in a restoration of the original coumarins (Lopez-Castillo N.N et al., 2013) (Fig. 2).
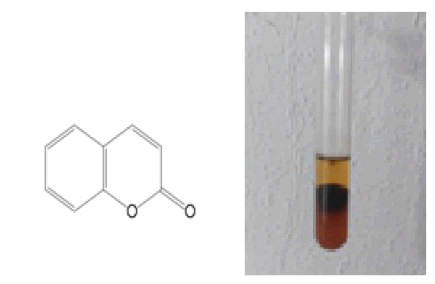
Figure 2: 2H-chromen-2-one.
Test for Anthocyanins
2ml of extract was treated with 2 ml of 2N hydrochloric acid and ammonia was added to it. The appearance of pink-red colour turning blue-violet. This indicates the presence of anthocyanin . Anthocyanin molecules will change their color depending upon the PH of their environment does it may serve as a PH indicator. The anthocyanin turns red to pink in acid (pH 16), (Fig.3) Reddish- purple in neutral solutions (pH 7) and green in alkaline or basic solution (pH 8-14) (Fossen et al., 1998).
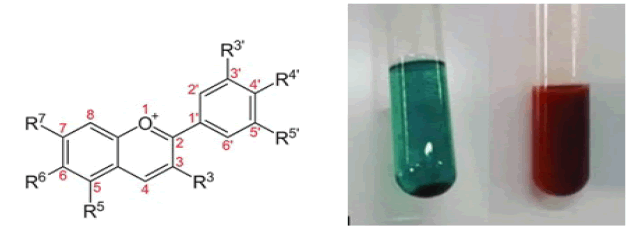
Figure 3: 3-galactoside.
Test for Leucoanthocyanins
5 ml of extract was allowed to react with 5 ml of isoamyl alcohol. Appearance of upper layer red in colour indicates the presence of leucoanthocyanins . They have no color of their own, but in acidic environment and at elevated temperature they are converted to colored anthocyanidis. This reaction is completion with condensation to a dimeric leucoanthocyanidi (Daneel F et al., 1999) (Fig.4).
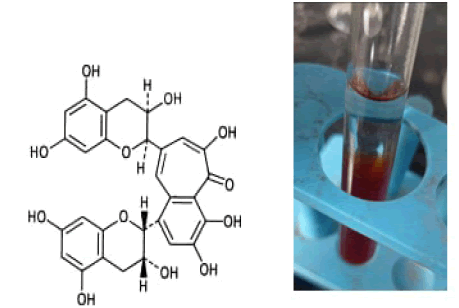
Figure 4: 2-Phenyl-3,4-dihydro-2H-1-benzopyran-3,4-diol
Test for Fatty acids
0.5 ml of extract was added to 5 ml of ether and allowed it to evaporate on filter paper. Then the filter paper was dried and the appearance of transparency on filter paper confirms the presence of fatty acids. The presence of these fatty acids in a considerable amount might serve to recognize the potential pharmacological importance of this plant in disease control (Aziz S et al., 2019) (Fig.5).

Figure 5: Test for steroids.
Test for Steroids
Libermann-Burchard Test: 1 ml of extract was dissolved in 10 ml of chloroform. To this mixture equal volume of concentrated sulfuric acid was added by sides of the test tube. The upper layer becomes red while lower layer of sulfuric acid turns yellow in colour with green fluorescence indicating the presence of steroids (Savithramma N. at al., 2011). Reactions of Steroids with acetic a hydride and sulphuric the libermann-Burchard Test R. It is then used to classify patient as having mild, classical or severe slows. The Libermann-Burchard or test anhydride testis used for the detection of cholesterol (Cook RP et al., 1961) (Fig.6).

Figure 6: 24-ethyl-lanostane.
Test for Saponins
Foam test: 2 ml of extract was taken in a test tube and 6 ml of distilled water was added to it. The mixture was then shaken vigorously. The persistence of foam was observed that indicates the presence of saponins (Savithramma N. at al., 2011). According to research saponins present in a given plant attribute form a soapy foaming substance when mixed with water persistent foam tests in acidic solutions, as well as blood hemolysis test are done to see the presence of saponins in a given mixture, as well as their capability to generate hemolysis (Hanz W et al., 2017) (Fig.7).
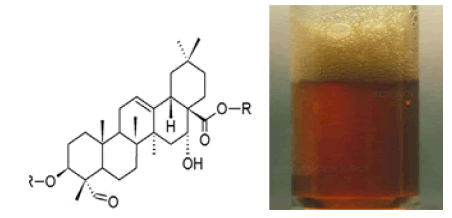
Figure 7: Triterpene glycosides.
Test for Terpenoids
Salkowski test: 2 ml of extract was treated with 2 ml of acetic anhydride. Few drops of concentrated sulfuric acid was then added to this solution and observed the formation of blue, green rings that indicates the presence of terpenoids (Savithramma N et al., 2011). Terpenoids can also be classified according to the number of cyclic structure they contain. The salkowski test can be used to identify the presence of terpenoids. At the least those containing an alcohol functional group, often arise by hydrolysis of carbocationic intermediates produced from geranyl Pyrophosphate (Fig.8).
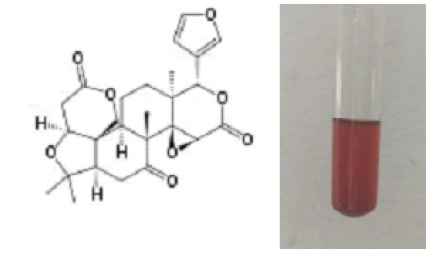
Figure 8: 7,16-Dioxo-7,16-dideoxylimondiol.
Test for Quinones
1 ml of extract was added to the 2 ml of dilute NaOH. Formation of blue green or red coloration confirms the presence of quinones (Soni A et al., 2013).On dilution a red color indophenol is formed which turns to deep blue color sodium salt solution of indophenol on treatment with sodium hydroxide (Fig.9).
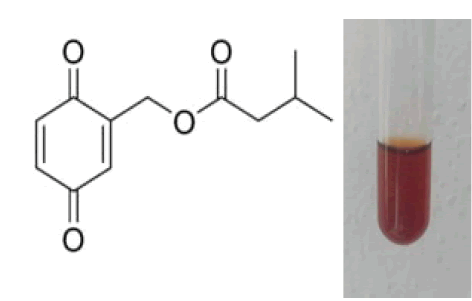
Figure 9: 1,4-benzoquinone or cyclohexadienedione.
Test for Tannins
Braymer’s test: 2 ml of extract was allowed to react with 10% alcoholic ferric chloride solution. Formation of blue or greenish colour of the solution was observed. This was the indication of the presence of the tannins (Soni A. et al., 2013). Hydrolysable tannins give bluish or precipitate and condensed tannins brownish to green ones. If the test is carried on and extract the contents both type of tannins, a blue color is produced which changed to olive green as more ferric chloride is added (Fig.10).
Figure 10: Hydrolyzable tannins.
Test for Phlobatannins
Precipitate test: About 2 ml of extract was added to 2 ml of 1% aqueous hydrochloric acid and the mixture was boiled. Deposition of a red precipitate confirmed the presence of phlobatannins (Soni A et al., 2013). Phlobatannis of precipitate test are found to be present in the least concentrations (Nathenial S et al., 2019) (Fig.11).
Figure 11: Tetrafucol A.
Test for Phenolic Compounds
Ferric chloride test: Few drops of the extract were treated with 5% aqueous ferric chloride. Formation of deep blue or black colour indicates the presences of phenolic compounds (Shah P et al., 2014). To detect the presence of a phenol functional group in a given sample. This test is based on the fact that the phenols a coloured complex with neutral ferric chloride solution (Fig.12).
Figure 12: Benzenol.
Test for Flavonoids
Alkaline reagent test: 2 ml of extract was treated with few drops of 1N sodium hydroxide solution and observed the formation of intense yellow colour. This yellow colour becomes colorless on addition of dilute hydrochloric acid, indicating the presence of flavonoids (Shah P et al., 2014). NaoH is completely ionic containing sodium cations and hydroxide anions. It is a sufficiently strong base which deprotonates phenol entirely. The purpose of 5% NaoH in this test was to deprotonates the polyphenolic molecules contained in flavonoids(Fig.13).
Figure 13: 2-phenyl-1,4-benzopyrone.
Test for Alkaloids
Mayer’s Test: 2 ml of extract was treated with 2 drops of Mayer’s reagent. Presence of white creamy precipitate indicates the positive test (Shah P et al., 2014). Most alkaloids are precipitated from neutral or slightly acidic solution by Mayer’s reagent (potentiomercuric iodide solution) to give a cream colored precipitate (Mayer’s. US Pharmacopeia. ,2012) (Fig.14).

Figure 14: phenethylamine alkaloid.
Determination of Ash content
2 g of each plant sample was taken and weighed accurately in a clean silica dish. The dish was first heated over a low burner flame. After that the dish is transferred to a muffle furnace maintained at 300°C-450°C for 15 min. The ash residue obtained was then cooled in desiccator and weighed (Motegaonkar M. B et al., 2012) (Fig. 15A-15C). The percentage of total ash content was calculated by the formula:

Figure 15: A. Unani herbal Medicine (B) Muffle furnace (C) Ash residue
Total Ash Percent of plant sample (%) = [Weight of dry ash residue (g) ÷ Weight of plant sample (g)] x 100 = 1.57/2 x 100 = 78.5%.
Determination of Ash Values
Total Ash: 3g of the test drug was accurately weighed and incinerated in a crucible dish at a temperature not exceeding 450°C until it was free from carbon. It was then cooled and weighed. The % w/w of ash with reference to the air-dried powder was calculated.
Water Soluble Ash: The total ash was obtained as the above method for preparation of total ash. The ash was boiled with 25 ml of water for 5mins. The insoluble ashes were collected using filter paper. It was then washed with hot water and transferred to the silica crucible. It was then ignited for 15minutes at temperature not exceeding 450°C. For determination of weight of the water soluble ash the silica
crucible and residue were weighed until constant weight was attained. The weight of the water soluble ash was determined by subtracting the weight of insoluble ash from the weight of total ash is 1.0 – 0.91 = 0.09(Fig.16A-16C).

Figure 16: (A) Boiling ash residue (B) Filtration (C) Collection of filtrate
Acid insoluble Ash: The total ash was obtained as the above method for preparation of total ash. The ash was boiled for 5 minutes with 25ml 10% Hcl. The insoluble ashes were collected using filter paper and washed with hot water. It was then transferred to the silica crucible and ignited for 15minutes at temperature not exceeding 450oC. The silica crucible and residue were weighed until constant weight is 1.09 – 0.39 = 0.61 (Fig.17).

Figure 17: (A) Boiling ash residue (B) Collection of filtrate
Determination of Extractive Value
Alcohol soluble extractive value: 3g of test drug powder was weighed and macerated with 100ml of ethanol in a closed container for 24 hours. The resulting solution was shaken continuously for 6 hours. It was then allowed to stand and soak for 18 hours(Fig.18A-18C).
Figure 18: (A) Concentration of extract (B) Filtration (C) Collection of filtrate
The solution was filtered and evaporated of the filtrate in a flat bottomed shallow dish and dried at 105°C. Then the content was cooled and weighed up to 1.34.
Water soluble Extractive value
3g of test drug powder was weighed and macerated with chloroform and water, respectively, at 80°C for 24 hrs. The resulting solution was shaken continuously for 6 hours and allowed to stand and soak for 24hrs then filtered. The solution from both chloroform and water respectively was filtered and evaporated of the filtrate in a flat bottomed shallow dish. It was dried at 105oC then cooled and weighed up to 0.33 (Fig.19A-19C).
Figure 19: (A) Boiling ash residue (B) Filtration (C) Collection of filtrate
Loss on Drying
The powdered drug was taken and dried in the oven at 100°C- 105°C to constant weight. The result was noted.
Physical characterization
Solubility: A little of the sample was shaken well with distilled water. A little of the sample was shaken well with Conc. Hcl and Conc. H2SO4. Sparingly soluble character indicates the presence of Silicate (Fig.20).
Figure 20: Sample with Conc HCL and Con H2SO4.
Action on heat: A small amount of the sample was taken in a dry test tube and heated gently. If there was a strong white fumes evolving it indicates the presence of Carbonate (Fig.21).
Figure 21: Action on heat
Flame test: A small amount of the sample was made into a paste with con. Hcl in a watch glass. It was then introduced into non-luminous part of the Bunsen flame. Appearance of bluish green flame indicates the presence of Copper (Fig.22).
Figure 22: Flame test
Ash Test: A filter paper was soaked into a mixture of sample and cobalt nitrate solution. It was then introduced into the Bunsen flame and ignited. Appearance of yellow colour flame indicates the presence of Sodium (Fig.23).
Figure 23: Ash Test
Bio-chemical analysis
Preliminary Basic and Acidic radical studies
Preparation of extract: 10 g of sample was taken in a 250 ml of clean beaker and 50 ml of distilled water was added to it. Then it was boiled well for about 10 mins. Then it was allowed to cool and filtered in a 100 ml volumetric flask and made up to 100 ml with distilled water. This preparation was used for the qualitative analysis of acidic/ basic radicals and biochemical constituents in it (Fig.24).
Figure 24: Sample was taken in a 250 ml.
Test for Basic radicals
Test for Potassium: To a pinch of the SPC 2 ml of sodium nitrate and 2 ml of cobalt nitrate solution in 30% glacial acetic acid was added and observed for the presence of yellow precipitate(Fig.25).
Figure 25: Test for Potassium
Test for Ammonium: To 2 ml of SPC extract few ml of Nessler's reagent and excess of sodium hydroxide solution are added for the appearance of brown colour (Fig.26).
Figure 26: Test for Ammonium.
Test for Iron (Ferrous): The SPC extract was treated with Conc. HNO3 and ammonium thiocyanate and waited for the appearance of blood red colour (Fig.27).
Figure 27: Test for Iron (Ferrous)
Test for Zinc: To 2 ml of the SPC extract drops of sodium hydroxide solution was added and observed for white precipitate formation(Fig.28).
Figure 28: Test for Zinc
Test for Lead: To 2 ml of SPC extract 2ml of potassium iodide solution was added and noted for yellow colored precipitate(Fig.29).
Figure 29: Test for Lead
Test for Copper : A pinch of SPC was made into a paste with con. Hcl in a watch glass and introduced into the nonluminous part of the flame and noted for blue colour appearance( Fig.30).
Figure 30: Test for Copper
Test for Mercury: To 2 m1 of the SPC extract sodium hydroxide solution was added and noted for yellow precipitate formation(Fig.31).
Figure 31: Test for Mercury
Test for Sulphate: To 2 ml of the SPC extract 5% of barium chloride solution was added and observed for the appearance of white precipitate (Fig.32).
Figure 32: Test for Sulphate
Test for Chloride: The SPC extract was treated with silver nitrate solution and observed for the appearance of white precipitate (Fig.33).
Figure 33: Test for Chloride
Test for Phosphate: The SPC extract was treated with ammonium molybdate and conc. HNO3 and observed for the appearance of yellow precipitate(Fig.34).
Figure 34: Test for Phosphate
Test for Carbonate: The SPC extract was treated with conc. Hcl and observed fourth appearance of effervescence (Fig. 35).
Figure 35: Test for Carbonate
Test for Fluoride & Oxalate: To 2ml of SPC extract 2ml of dil.acetic acid and 2ml calcium chloride solution was added and heated and watched for cloudy appearance(Fig. 36).
Figure 36: Test for Fluoride & Oxalate
Test for Nitrate: To 1 gm of the SPC, copper turnings was added and again conc.H2SO4 was added, heated and the test tube was tilted vertically down and observed for yellowish red colour(FIG. 37).
Figure 37: Test for Nitrate
Results and Discussion
ve a significant proportion of key for phytochemicals that can be recognized by tests of quality. In our analysis it was cleared that the Niswani Tea is rich in alkaloids, flavonoids, saponins, tannins, fatty acids, quinones, coumarins, leucoanthocyanins, steroids etc., The important thing is that all plant samples contain one common and abundant secondary metabolite, flavonoid. From the literature survey it was found that flavonoids have wide range of biological properties such as anti-inflammatory, antibacterial, antiviral, anti-allergic, cytotoxic antitumor properties. It is used in the treatment of neurodegenerative diseases and has vasodilatory action. It is also reported that flavonoids involved in inhibition of lipid peroxidation, platelet aggregation, capillary permeability and fragility, cyclo-oxygenase and lipoxygenase enzyme activities etc. Flavonoids are also known to inhibit variety of enzymes like hydrolases, hyaluronidases, alkaline phosphatases, arylsulphatases, cAMP phosphodiesterases, lipase, á´¼- glucosidase and kinases. According to the literature, it also has been found that quercetin, plays an important role in diabetes. It leads to the regeneration of pancreatic islets and increases insulin release. It also stimulates Ca2+ (Sandhar H.K et al., 2011).Uptake from isolated islet cells which is helpful in non-insulin dependent diabetes.
Alkaloids and phenolic compounds along with hypoglycemic (Tiong S. H et al., 2013 ; Maiti D et al., 2012), antidiabetic properties also exhibit anti-inflammatory, antimicrobial and antioxidant effects (Al-Shahwany A. W et al., 2014). Moreover, saponins exhibit various biological activities like it gives a permeability to the cell membrane, helpful in lowering the serum cholesterol levels, it possesses abortifacient properties, it has immunomodulatory property, it has cytotoxic effects on malignant tumor cells and is involved in synergistic enhancement of the toxicity of immunotoxins (Thakur M et al., 2011). Saponins also show antidiabetic property (Zheng T et al., 2012).
Coumarins, a major class of flavonoids, possesses pharmacological properties like antidiabetic, antioxidant, hepato-protective, anticoagulant, antimicrobial, anti-inflammatory, Analgesic, antioxidant, anticancer, antiviral, antimalarial activities etc (Sahoo S. S et al., 2012). Tannins are reported to have a cardio-protective, anti-inflammatory, anticarcinogenic and antimutagenic properties. Tannins are also involved in treatment of noninsulin dependent diabetes mellitus by enhancing the glucose uptake and inhibiting abiogenesis (Kumari M et al., 2012). The most striking feature about quinones is its pharmacological properties that makes it different from other secondary metabolites. It inhibits HIV 1 reverse transcript (Valderrama J. A et al.,2003) and shows antitumor and immunomodulatory activities. It also has antimicrobial, anticancer, antiviral and antibacterial properties (Masih D et al., 2019). The main purpose to calculate the total ash content is to measure the total amount of minerals present in that plant samples. Among the four plants tested for total ash content, Niswani Tea shows highest percentage of the ash i.e. it contains 78.5% of ash indicating high amount of minerals in it. Minerals are used as coenzymes and cofactors in the biochemical processes. Therefore, it is necessary to evaluate the mineral values in the extract.
The results of our investigation proved the presences of various phytochemicals such as coumarins, anthocyanins, leucoanthocyanins, fatty acids, steroids, saponins, terpenoids, quinones, tannins, phlobatannins, phenolic compounds, flavonoids, alkaloids and determination of ash content in the Niswani Tea which may be responsible for pharmacological action.
This medicine is used in the treatment of several health related problems in the production of medicines for a number of infectious diseases, including chronic ulcers, leucorrhoea, pyorrhea and fungal infections of the skin. It is good for girl’s/women care, formulated with ashoka and kalongi Berg-e-Heena and Gull-e-Nilofer, it is very effective caffeine free herbal remedies for Heart Care and Blood pressure.
As herbal medicine is more affordable than conventional medicine, natural healing, strength in immune system, fewer side effects, cost effective, stabilizes hormones, metabolism, easier to obtain than prescription medicine.
Conclusion
The unani herbal medicine researched were determined to be high in phytochemicals are full of pharmacological and therapeutic value. Flavonoid is abundant in almost all plant species investigated in making it the most abundant secondary metabolites. Further research is needed to determine their potential in the biological qualities listed above, such as antidiabetic and anti-tumor properties among others. Saponins and tannins are also present in almost all species studied. It was concluded that the herbal medicine extraction was rich in phytochemicals with significant pharmacological and medicinal applications. Utilizing knowledge of phytochemical constituents present in unani herbal medicine drugs and its consideration in therapeutics can improve the patient care and can contribute further in global requirement of traditional health care facility by Unani herbal Medicine.
References
"Ferric Chloride — Pyridine Test Page". Chemistry.ccsu.edu. Retrieved 2013-09-11. [Google Scholar] [Cross Ref]
14 December (2022). Creative Commons Attribution-ShareAlike License 3.0. [Google Scholar] [Cross Ref]
Al-Shahwany A. W. (2014). Alkaloids and Phenolic Compound Activity of Piper Nigrum against Some Human Pathogenic Bacteria. Biomed. Biotech. 2: 20-28 [Google Scholar] [Cross Ref]
Ashok P.K., Upadhyaya K. (2012). Tannins are Astringent. J Pharm. Phyto. 1: 45-50. [Google Scholar] [Cross Ref]
Aziz S., Mitu T.K. (2019). Analysis of fatty acid and determination of total protein and phytochemical content of Cassia sophera Linn leaf, stem, flower, and seed. Beni-Suef Univ J Basic Appl. Sci. 8: 1-9. [Google Scholar] [Cross Ref]
Balandrin M.F., Klocke J.A., Wurtele E.S., Bollinger W. H. (1985). Natural plant chemicals: Sources of industrial and medicinal materials. Sci. 228: 1154-1160. [Google Scholar] [Cross Ref]
Bansode T.S., Salalkar B.K. (2015). Phytochemical analysis of some selected Indian medicinal plants. Int J Pharm Bio Sci. 6: 550-556. [Google Scholar] [Cross Ref]
Cook RP. (1961). Reactions of steroids with acetic anhydride and sulphuric acid (the Liebermann-Burchard test). Analyst. 86: 373-81. [Google Scholar] [Cross Ref]
Daneel F., Riaan B. (1999). How are leucoanthocyanidins converted to colored compounds? - Compr Nat Prod Chem Bing [Google Scholar] [Cross Ref]
Fossen. (1998). How does anthocyanin work as a pH indicator? - Studybuff [Google Scholar] [Cross Ref]
Gracelin D.H., Britto A., Kumar B.J. (2013). Qualitative and quantitative analysis of phytochemicals in five Pteris species. Int J Pharm Pharm Sci. 5: 105-7. [Google Scholar] [Cross Ref]
Gul R., Jan S.U., Faridullah S., Sherani S., Jahan N. (2017). Preliminary phytochemical screening, quantitative analysis of alkaloids, and antioxidant activity of crude plant extracts from Ephedra intermedia indigenous to Balochistan. Sci World J. 13: Article ID 5873648. [Google Scholar] [Cross Ref]
Hanz W. (2017). Test for Saponins. HomeSteady. [Google Scholar] [Cross Ref]
Hecker E., Kinghorn A. D. (1977). Toxic Plants, Ed., Pure Appl. Chem. 49, 1243. [Google Scholar] [Cross Ref]
Hussain M.S, Fareed S., Ansari S., Rahman M.A., Ahmad I.Z., Saeed M. (2012). Current approaches toward production of secondary plant metabolites. J pharm bioallied sci. 4: 10. [Google Scholar] [Cross Ref]
Kumari M., Jain S. (2012). Tannins: An Antinutrient with Positive Effect to Manage Diabetes. Res. J Recent Sci. 1: 70-73. [Google Scholar] [Cross Ref]
Lopez-Castillo N.N., Rojas-Rodriguez A.D., Porta B.M., Cruz-Gomez M.J. (2013). Process for the obtention of coumaric acid from coumarin: analysis of the reaction conditions. 3: 195-201. [Google Scholar] [Cross Ref]
Maiti D., Majumdar M. (2012). Impact of bioprocessing on phenolic content and antioxidant activity of soy seed to improve hypoglycemic functionality. Asian J Plant Sci. Res. 2: 102-109. [Google Scholar] [Cross Ref]
Masih D, Singh N, Singh A. (2019). Red beetroot: A source of natural colourant and antioxidants: A review. J Pharmacogn Phytochem. 8: 162-166. [Google Scholar] [Cross Ref]
Modi D.C., Patel J.K., Shah B.N., Nayak B.S. (2010). Pharmacognostic studies of the seed of Syzygium cumini Linn. Pharma Sci. Monit. 1: 20-6. [Google Scholar] [Cross Ref]
Motegaonkar M. B., Salunke S. D. (2012). The Ash and Iron Content of Common Vegetable Grown in Latur District, India. Res J Recent Sci. 1: 60-63, [Google Scholar] [Cross Ref]
Nathenial S., Fatima A., Fatima R., Ijaz N., Saeed N., Shafqat A., Leghari L. (2019). Phytochemical study of acetone solvent extract of Coriander sativum. J Pharmacogn Phytochem. 8: 136-40. [Google Scholar] [Cross Ref]
Priya , SCRIBD , Article No: 355653848 Tests For Flavonoids | PDF | Sodium Hydroxide | Alkene (scribd.com) [Google Scholar] [Cross Ref]
Sahoo S. S., Shukla S., Nandy S., Sahoo H. B. (2012). Synthesis of novel coumarin derivatives and its biological evaluations. Euro. J. Exp. Bio. 2: 899-908, [Google Scholar] [Cross Ref]
Sandhar H.K., Kumar B., Prasher S., Tiwari P., Salhan M., Sharma P. (2011). A Review of Phytochemistry and Pharmacology of Flavonoids. Int Pharma Sci. 1: 1. [Google Scholar] [Cross Ref]
Savithramma N., Linga Rao M., Suhrulatha D. (2011). Screening of Medicinal Plants for Secondary Metabolites. Middle-East J Sc. Res. 8: 579-584, [Google Scholar] [Cross Ref]
Shah P., Modi H. A., Shukla M. D., Lahiri S. K. (2014). Preliminary Phytochemical Analysis and Antibacterial Activity of Ganoderma lucidum collected from Dang District of Gujarat, India. Int J Curr Microbiol App Sci. 3: 246-255. [Google Scholar] [Cross Ref]
Sigma Aldrich. Retrieved 2012-02-08, US Pharmacopeia. Retrieved 2012-02-08. Mayer's reagent - Wikipedia [Google Scholar] [Cross Ref]
Soni A., Sosa S. (2013). Phytochemical analysis and free radical scavenging potential of herbal and medicinal plant extracts. J Pharmacog phytochem. 2: 22-9. [Google Scholar] [Cross Ref]
Test for Phenolic Group - Chemistry Practicals Class 12 (byjus.com) Wikipedia · heimduo.org Wikipedia text under CC-BY-SA license [Google Scholar] [Cross Ref]
Thakur M., Melzig M. F., Fuchs H., Weng A. (2011). Chemistry and pharmacology of saponins: Special focus on cytotoxic properties. Bot.: Targets Ther. 1: 19-29, [Google Scholar] [Cross Ref]
Tiong S. H., Looi C. Y., Hazni H., Arya A., Paydar M., Wong W. F., Cheah S. C., Mustafa M. R., Awang K. (2013). Antidiabetic and Antioxidant Properties of Alkaloids from Catharanthus roseus (L.) G. Don. Mol. 18: 9770-9784, [Google Scholar] [Cross Ref]
Tiwari P., Kumar B., Kaur M., Kaur G., Kaur H. (2011). Phytochemical screening and extraction: a review. Int pharm sci. 1: 98-106. [Google Scholar] [Cross Ref]
Ugochukwu S.C., Uche A., Ifeanyi O. (2013). Preliminary phytochemical screening of different solvent extracts of stem bark and roots of Dennetia tripetala G. Baker. Asian j plant sci res. 3:10-13. [Google Scholar] [Cross Ref]
Valderrama J.A., Benites J., Cortes M., Pessoa-Mahana H., Prina E., Fournet A. (2003). Studies on Quinones. Part 38: synthesis and leishmanicidal activity of sesquiterpene 1, 4-Quinones. Bioorg. Med. Chem. 3; 11: 4713-4718, [Google Scholar] [Cross Ref]
Zheng T., Shu G., Yang Z., Mo S., Zhao Y., Mei Z. (2012). Antidiabetic effect of total saponins from Entada phaseoloides (L.) Merr. in type 2 diabetic rats. J Ethnopharmacol. 139: 814-821. [Google Scholar] [Cross Ref]