Research - Modern Phytomorphology ( 2020) Volume 14, Issue 2
Most recent detection of invasive species Erysiphe palczewskii (Jacz.) U Braun et S Takam on Robinia pseudoacacia L. in Ukraine
Klіuchevych MM1, Stoliar SH1*, Chumak PYa1, Retman SV2, Strygun OO2, Tkalenko HM2 and Vigera SM12Institute for Plant Protection of NAAS of Ukraine, 33 Vasylkivska Street, 03022, Ukraine
Stoliar SH, Zhytomyr National Agroecological University, Staryi Blvd 7, Zhytomyr, 10008, Ukraine, Email: svetlana-stolyar@ukr.net
Received: 24-Apr-2020 Accepted: 10-Aug-2020 Published: 18-Aug-2020, DOI: 10.5281/zenodo.5077865
Abstract
The investigation of morphological traits of main development cycles of Erysiphe palczewsk Takamatsuii on various host plant species at seven different ecological geo-points in Ukraine. Sampling was conducted with the help of plant route research method in the parks in Kiev and Kiev region, and also in Zhytomyr. Phytopathologic analysis, morphometry of conidia, fruit bodies and their appendages as well as asci and ascospores were carried out using a light microscope and a smartphone with the software “Magnifier Cam”. The greatest disease development is manifested on the fruits (beans), leaves, branches of current annual accretion on Robinia pseudoacacia L. For the first time in Ukraine, all development stages of invasive species Erysiphe palczewskii were detected on black locust (Robinia pseudoacacia L.). Comparative analysis of morphometric parameters of conidia, fruit bodies, appendages, asci and ascospores from Caragana arborescens and black locust from different ecological geo-points was conducted.
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Haberler
Keywords
powdery mildew, Erysiphe palczewskii, black locust, Caragana arborescens, norm of reaction
Introduction
Fungi belonging to the genus Erysiphe DC (Erysiphales, Erysiphaceae) are widespread throughout the world and cause diseases of powdery mildew on numerous herbaceous and woody plants of the class Magnoliopsida. Many of these fungi are cosmopolitic (Braun et al. 2009). According to Takamatsu et al. (2015), the genus Erysiphe is considered to be the largest of Erysiphaceae and comprises more than 50% of all species in this family.
Erisyphaceae have recently attracted the attention of many researchers in connection with the emerging tendency to a high expansion and pathogenicity of some species. For example, in England from 1970 to 2004, more than 200 phytopathogenic organisms were registered for the first time, including 184 fungi, 26 viruses, 23 bacteria and one phytoplasma. Among the species of Erisyphaceae of the genus Erysiphe on the Siberian pea shrub (Caragana arborescens Lam.), invasive species E. palczewskii (Jacz.) U. Braun et S. Takam. was also detected (Jones & Baker, 2007). According to V. P. Geliuta (Geliuta, 1989), the invasive species E. palczewskii used to be spread only on the Siberian pea shrub in China, the Russian Federation (in the Far East and Siberia), Kazakhstan, Turkmenistan, in Poland, in Ukraine.
Research on the issue of the current distribution of the invasive species E. palczewskii indicates that the fungus was detected in many European countries (Austria, England, Belarus, Germany, Hungary, Spain, Latvia, Lithuania, Poland, Romania, Slovakia, Finland, Sweden, Czech Republic), the USA and Canada (Brown 1995; Huhtinen 2001; Nischwitz & Newcombe 2003; Glawe & Laursen 2005; Glawe et.al. 2006; Jones & Baker 2007; Lebeda et al. 2008).
In the process of the expansion of E. palczewskii to other continents there was an extension of the phylogenetic host specialisation of the fungus. Out of the 80–100 species of the genus Caragana Lam. (Fabaceae) in addition to C. arborescens the pathogen was detected on more than 10 species, as well as on Robinia pseudoacacia L. While collecting fungal fruit from R. pseudoacacia, apart from bipalmate apices a high occurrence of trichotomy was noted. This encouraged U. Brown to describe a new invasive species Microsphaera subtrichotomy. However, as V.P. Geiluta (Geliuta, 1989) notes, this feature is characteristic also of apices E. palczewskii from the plants of the genus Caragana. Therefore, this author considers M. subtrichotoma to be a synonym of E. palczewskii.
Owing to the fact that it is difficult to identify the described Erisyphaceae on Robinia pseudoacacia, U. Brown and his co-authors (Braun et al. 2009) revised the descriptions of these species and created a key for their identification. As a result, it was established that currently there are only four invasive species on Robinia pseudoacacia: Erysiphe trifolii, Erysiphe palczewskii, Erysiphe pseudacaciae and Erysiphe robiniicola (Braun et al., 2009). Studies of the genome Erysiphe palczewskii and of other species of the complex from Robinia pseudoacacia confirmed the independence of this species (Braun et al. 2009; Lee & Nguyen 2018).
It should be noted that the invasive species E. palczewskii has been studied in Ukraine for quite a long time and the fungus is well studied on C. arborescens. However, it was detected only once on Robinia pseudoacacia in Lutsk (Geliuta 1989). And thirty years later, several reports appeared about the detection of E. palczewskii on Robinia pseudoacacia in China (Braun et al. 2009) and Korea (Lee & Nguyen 2018).
The purpose of the research is to carry out a more thorough study of the anamorphic and teleomorphic stages of the invasive species E. palczewskii and to conduct a comparative description of the morphological features of the fungus from the nutritive plants C. arborescens and R. Ñ?seudoacacia from different ecological and geographical points of Ukraine and other countries.
Materials and Methods
Samples of powdery mildew for the research were selected with the help of plant route research method in the parks of Kiev: “Kin Grust” (50.521017 N, 30.444883 E), “Natalka” (50.50655 N, 30.513758 E), in the V. Chkalov garden square (50.450055 N.; 30.502865 E), in the A. V. Fomin Botanical Garden (BsF) (50.444958 N; 30.50236 E). In addition, black locust was examined in village Glubokoe, Borispol Area, Kiev Region (50.265193 N, 30.92781 E). In Zhytomyr, powdery mildew was discovered in the Botanical Garden of the Zhytomyr National Agroecological University (BsZhNAEU) (50.252043 N, 28.696878 E) and in the Yu. Gagarin park (50.24521 N, 28.664937 E). Freshly selected samples of powdery mildew were used for the research. The samples comprised not less than 300 specimens of conidia and fungal fruit from the populations under investigation. Morphometry of conidia, fungal fruit and their appendages, as well as sacs and ascospores was carried out using a microscope with an objective micrometer and a smartphone with the software “Magnifier Cam”. A number of parameters were studied for the estimation of the variability and interconnection of the morphological features of the fungus in different types of nutritive plants at different ecological and geographical points. They include length and width of the conidia, the diameter of the fungal fruit, the number and length of the appendages, the number of sacs and ascospores. The range of reaction rates of the signs was determined using the formula:
D = a – c,
where D-the range of reaction rates;
◊-maximum (max) indicator of attribute parameters
◊–minimum (min) indicator of attribute parameters
The phytopathological analysis of the samples was carried out according to the method of V.P. Geliuta (1989); S.V. Shevchenko; A.V. Tsyliurik (1986). Powdery mildew was identified using the keys (Geliuta 1989; Braun et al. 2009). Statistical data processing was performed using Microsoft Exel 7.0 and the STATISTICA-Apreadsheet.
Results
Powdery mildew Erysiphe palczewskii (Jacz.) U. Braun and S. Takam. is widespread on the Caragana arborescens in the garden squares, parks and botanical gardens we investigated in Kiev and Zhytomyr. Less often, phytopathogen was detected on black locust: Park “Kin Grust”, village Glubokoie and Botanical Garden of Zhytomyr National Agroecological University (Tab. 1.).
| Place of detection | Host plant | Developmental cycle | ||
| City, village | Coordinates, park, garden square | Anamorph | Telemoprh | |
| Kyiv | 50.444958 N; 30.50236 E, BsF | Caragana arborescens | + | + |
| 50.450055 N; 30.502865 E, V. Chkalov garden square | Caragana arborescens | + | + | |
| 50.50655 N, 30.513757 E, park “Natalka” | Caragana arborescens | + | + | |
| 50.521017 N, 30.444383 E, park “Kin Grust” | Robinia pseudoacacia | + | _ | |
| Village Glubokoie, Boryspol raion, Kyiv region | 50.265193 N, 30.927810 E., orchard | Robinia pseudoacacia | + | + |
| Zhytomyr | 50.252043 N, 28.696878 E, BsZhNAEU | Robinia pseudoacacia | + | + |
| 50.24521 N, 28.664937 E, Yu. Gagarin park | Caragana arborescens | + | + | |
Table 1. Location of the investigated samples of powdery mildew Erysiphe palczewskii on nutritive plants at different ecological and geographical points (2016–2018).
It is known (Geliuta 1989) that the Erysiphe (Microsphaera) palczewskii on Caragana arborescens is characterized by white or dingy white powdery mycelium. It can be dense and well developed. The anamorph is of Pseudoidium type. Conidia are cylindrical, rounded at the ends, 21-37 x 11–19 microns. Cleistothecium is in huge quantities; it completely covers the affected organs; it is dark brown, semi-globular, 100–143 microns in diameter. Appendages of the second type are in the lower part of the fungal fruit or equatorial; they extend away almost vertically; they are not numerous (upto 16), long (152–363 microns), colourless. Apices are 4–7 fold bipalmate: branches are finite and usually straight. The characteristic feature of apices is the high occurrence of trichotomy in branching of various orders, which is the result of the disorder in dichotomy as well as the presence of the main axes in their finite parts, which are formed in consequence of the transformation of dichotomous branching into a false-monopodial one. Sacs are ellipsoidal, inequilateral; there are 7–10 of them; they have short legs that are easy to break; the size is 53–82 x 27–39 microns; predominantly they have 5–6 spores. Spores are ellipsoidal, elongated ovoid; the size is 17–27 x 10–16 microns.
The fungus forms a white surface mycelium and brown necrosis on the top or bottom of the leaves on Robinia pseudoacacia (Lee & Nguyen 2018). Conidiophores are cylindrical, 43.5–68.5 x 6.5–8.5 microns. Conidia are formed separately; they vary from ellipsoidal to cylindrical, 27.5–40.5 x 13.0–16.5 microns. Cleistothecium is 85.5–122.5 microns in diameter, spherical, dark brown with bipalmate apices; the upper surfaces of leaves are most densely covered with it. Fungal fruit had 2–8 sprays, 113–245.5 microns in length, 6.5–9 microns in width, 0–1 septum. Sacs 51–64.5 x 30.5–37.5 microns with 2–6 ascospores. Spores are ellipsoidal, 18.5–23.5 x 9.0–12.5 microns.
Thus, on various species of nutritive plants at different ecological and geographical points, the morphological characteristics of the invasive species E. palczewskii (syn. Microsphaera palczewskii) differs markedly both in terms of the length and width of the conidia, the diameter of the fungal fruit, the number of appendages, the dimensions of the sacs and the spores in them.
The investigated samples of powdery mildew from the host plant of Siberian pea shrub were characterized both by the anamorphic and telemorphic cycle of development. For the first time the formation of fungal fruit on fruits (beans) was detected on black locust. In some cases the fungus had a complete cycle of development (conidia and cleistothecium) (village Glubokoie and BsZhNAEU), and sometimes the telemorphic cycle of development was not observed throughout the whole vegetation period of development of a host plant (park “Kin Grust”).
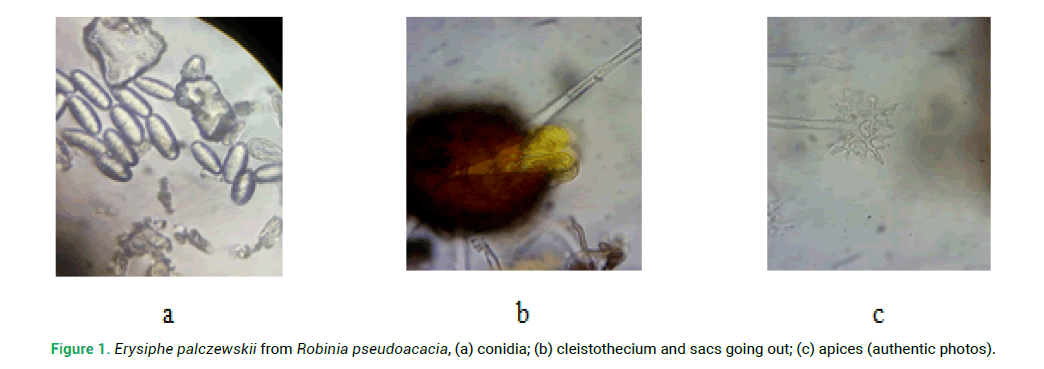
Measurements of the parameters of conidia Erysiphe palczewskii from different species of nutritive plants showed that on Caragana arborescens their length was 22–36 microns, width 10–18 microns, on Colutea arborescens 30.5–45.5 and 13.5–20.5, respectively, and on Robinia pseudoacacia, 21–37 and 12–18 microns respectively (Tab. 2.) and (Fig. 1a.).
Figure 1: Erysiphe palczewskii from Robinia pseudoacacia, (а) conidia; (b) cleistothecium and sacs going out; (c) apices (authentic photos).
| Author | Length, µm | Width, µm |
| Caragana arborescens Lam. | ||
| Braun (1995) | 21–37 | 11–19 |
| Geliuta (1989) | 21–37 | 11–19 |
| Glawe et al. (2006) | 26–32 | 11–15.5 |
| Lebeda et al. (2008) | 20–37.5 | 8.5–17.5 |
| Lonergan and Skoglund (2013) | 25–32 | 7–12 |
| Vajna (2006) | 26–40 | 15–18 |
| Authors | 22–36 | 10–18 |
| Colutea arborescens L. | ||
| Schmidt and Scholler (2002) | 30.5–45.5 | 13.5–20.5 |
| Robinia pseudoacacia L. | ||
| Lee and Nguyen (2018) | 27.5–40.5 | 13.0–16.5 |
| Authors | 21–37 | 12–18 |
Table 2. Morphometric parameters of conidia of Erysiphe palczewskii on various species of nutritive plants according to different researchers.
Measurements of the parameters of the fungal fruit and the appendages of the invasive species Erysiphe palczewskii showed that the fruit bodies on Caragana arborescens are 80–145 microns in diameter (Fig. 1b.) the appendages are not numerous 6–12 and 170–295 microns in length. On Robinia pseudoacacia, the fungal fruit is 73–123 microns, there are 6–10 appendages and their length is 105–280 microns (Tab. 3.).
| Author | Diameter of fungal fruit, µm | Appendages | |
| number | length, µm | ||
| Caragana arborescens Lam. | |||
| Braun (1995) | 80–140 | 5–12 | – |
| Geliuta (1989) | 100–143 | Upto 16 | 152–363 |
| Glawe et al. (2006) | 100–130 | – | 145–290 |
| Lebeda et al. (2008) | 67.5–112.5 | 3–13 | 180–330 |
| Lonergan and Skoglund (2013) | 100–122 | 5–10 | – |
| Vajna (2006) | 90–160 | 9–12 | 230–312 |
| Authors | 80–145 | 6–12 | 170–295 |
| Colutea arborescens L. | |||
| Schmidt and Scholler (2002) | 86–150 | 9–12 | – |
| Robinia pseudoacacia L. | |||
| Lee and Nguyen (2018) | 85.5–122.5 | 2–8 | 113–245.5 |
| Authors | 73–123 | 6–10 | 105–280 |
Table 3. Morphometric parameters of the fungal fruit and the appendages of Erysiphe palczewskii on various species of nutritive plants according to different researchers.
The number of sacs in the fungal fruit and the number of ascospores as well as morphometric parameters of the invasive species Erysiphe palczewskii taken from different species of host plants of the fungus are shown in Tab. 4.
| Author | Sacs | Ascospores | ||||||
| number | length, µm | width, µm | number | length, µm | width, µm | |||
| Caragana arborescens Lam. | ||||||||
| Braun (1995) | 45–80 | 25–40 | 14–27 | 8–15 | ||||
| Geliuta (1989) | 7–10 | 53–82 | 27–39 | 7-10 | 17–27 | 10–16 | ||
| Glawe et al. (2006) | 51–67 | 29–43 | 6–8 | 17.5–23 | 10–13.5 | |||
| Lebeda et al. (2008) | 47–77.5 | 22–42 | – | 17–27 | 8–16 | |||
| Lonergan and Skoglund (2013) | 48–54 | 34–43 | 3–6 | 14–17 | 9-11 | |||
| Vajna (2006) | 6–10 | 4.6–8 | 20–28 | 10–18 | ||||
| Authors | 6–10 | 50–59 | 24–40 | 5–8 | 16–25 | 10–15 | ||
| Colutea arborescens L. | ||||||||
| Schmidt and Scholler (2002) | 6–13 | 4–6 | 17–20 | 10–12 | ||||
| Robinia pseudoacacia L. | ||||||||
| Lee and Nguyen (2018) | 51–64.5 | 3.5–37.5 | 2–6 | 18.5–23.5 | 9.0–12.5 | |||
| Authors | 5–7 | 35–45 | 28–35 | 4–6 | 14–20 | 9–11 | ||
Table 4. Morphometric parameters of sacs and ascospores of the invasive species Erysiphe palczewskii on various species of nutritive plants according to different researchers.
Discussion
After the analysis of the samples of powdery mildew collected from nutritive plants of Caragana arborescens and plants investigated by other authors, it appears that in most cases conidia are ellipsoidal (ratio of length to width 1.9–2.1) and only in samples from the USA of North Dakota (Glawe et al. 2006) and Montana (Lonergan and Skoglund 2013) conidia are cylindrical (ratio 3.0). Specimens of conidia from Colutea arborescens and Robinia pseudoacacia tended to vary from ellipsoidal (authors, the ratio 1.9) to cylindrical; ratio 2.2–2.3 (Schmidt and Scholler 2002; Lee and Nguyen 2018).
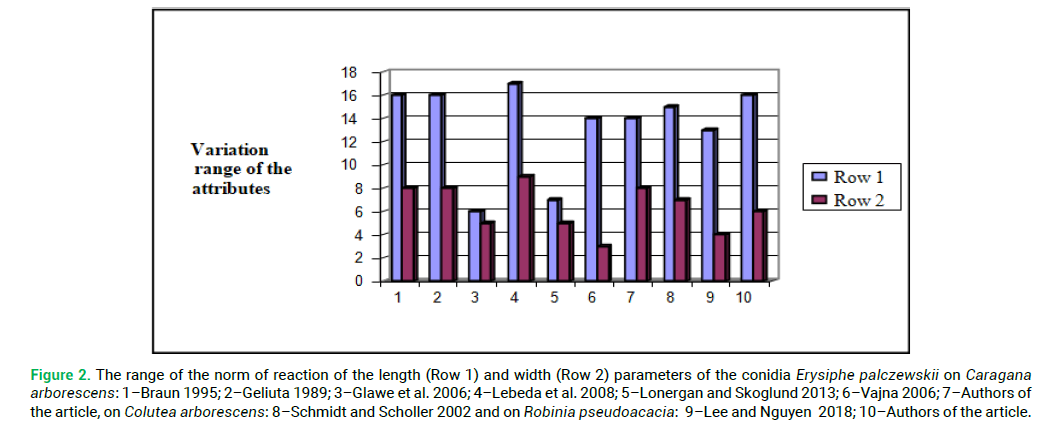
Each species of Erisyphaceae is characterized by its specific variation range of the parameters of the attributes (Chumak et al. 2012; Palagecha and Chumak 2011). Therefore, there is a need to analyse the peculiarities of the norms of reaction of the length and width parameters of the conidia of the invasive species Erysiphe palczewskii from various nutritive plants Tab. 5 and Fig. 2.
| Author | Length, µm | Width, µm |
| Caragana arborescens Lam. | ||
| Braun (1995) | 16 | 8 |
| Geliuta (1989) | 16 | 8 |
| Glawe et al. (2006) | 6 | 4 |
| Lebeda et al. (2008) | 17 | 9 |
| Lonergan and Skoglund (2013) | 7 | 5 |
| Vajna (2006) | 14 | 3 |
| Authors | 14 | 8 |
| Colutea arborescens L. | ||
| Schmid and Scholler (2002) | 15 | 7 |
| Robinia pseudoacacia L. | ||
| Lee Nguyen (2018) | 13 | 3 |
| Authors | 16 | 6 |
Table 5. Norm of reaction of the parameters of the conidia of invasive species Erysiphe palczewskii on various species of nutritive plants according to different researchers.
Figure 2: The range of the norm of reaction of the length (Row 1) and width (Row 2) parameters of the conidia Erysiphe palczewskii on Caragana arborescens: 1–Braun 1995; 2–Geliuta 1989; 3–Glawe et al. 2006; 4–Lebeda et al. 2008; 5–Lonergan and Skoglund 2013; 6–Vajna 2006; 7–Authors of the article, on Colutea arborescens: 8–Schmidt and Scholler 2002 and on Robinia pseudoacacia: 9–Lee and Nguyen 2018; 10–Authors of the article.
It should be noted that the norm of reaction of conidia of powdery mildew on different types of nutritive plants in different ecological and geographical points is not the same. The greatest differences in the range of length and width parameters of conidia are found between the cenopopulations of Caragana arborescens, which grows in North America (Glawe et al. 2006; Lonergan and Skoglund 2013) and the cenopopulations in Europe (Braun 1995; Geliuta 1989; Lebeda et al. 2008; Vajna 2006).
The cenopopulations of the fungus from Colutea arborescens and Robinia pseudoacacia are almost identical in terms of the range of the norm of reaction of the attributes of conidia (length and width).
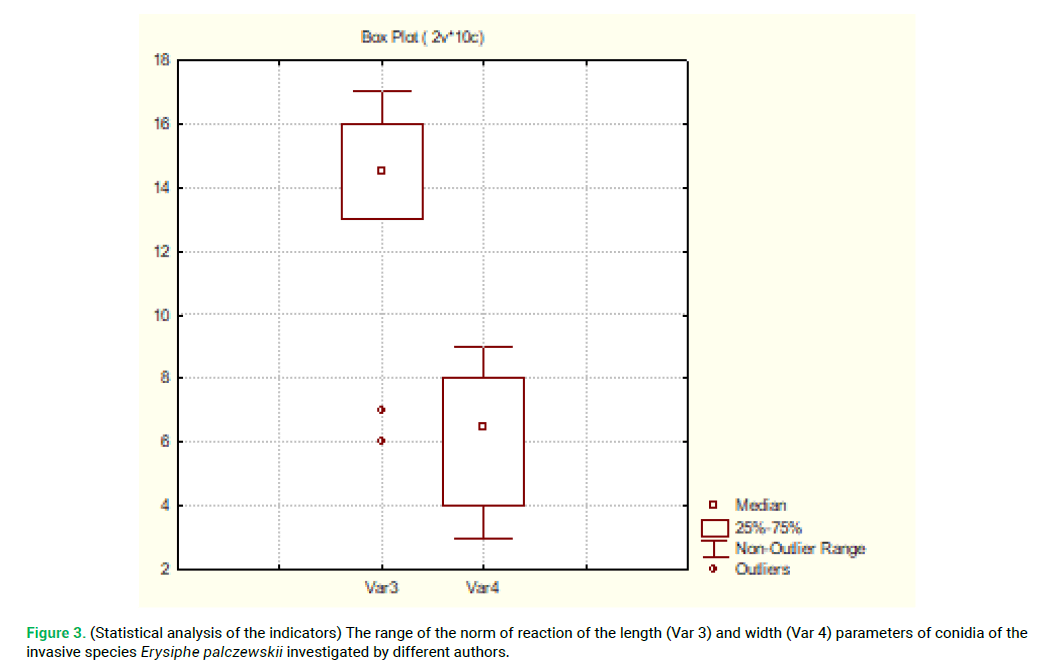
The conducted statistical analysis of the data from Tab. 5 is presented in Fig. 3.
Figure 3: (Statistical analysis of the indicators) The range of the norm of reaction of the length (Var 3) and width (Var 4) parameters of conidia of the invasive species Erysiphe palczewskii investigated by different authors.
The statistical analysis of the indicators of the range of the norm of reaction of the length (Var3) and width (Var4) parameters of conidia of the invasive species Erysiphe palczewskii investigated by different authors shows that only the data of the variation range of the length of conidia of the fungus from the American continent stand out significantly among other data provided (two points in Fig. 3.).
Thus, under the stressful conditions related to the invasion in new living environment, the variation range of the length parameters of conidia of Erysiphe palczewskii varies significantly, which is to be expected due to the decrease in the variability of the gene pool of the fungus.
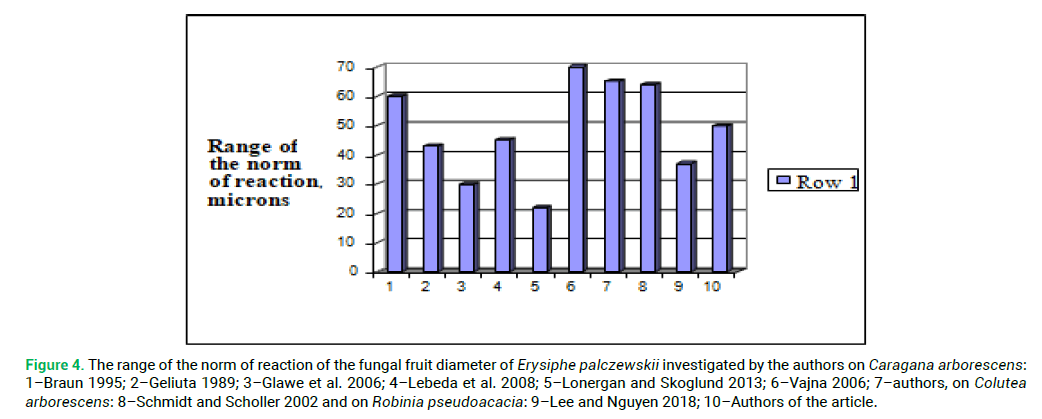
Analysis of morphometric parameters of cleistothecium and appendages of the invasive species Erysiphe palczewskii on different types of nutritive plants at different ecological and geographical points showed that the norm of reaction of the diameter of the fungal fruit of powdery mildew, the number of appendages and their length are not the same (Tab. 6.). The greatest differences in the range of indicators of the fungal fruit diameters are noted between the cenopopulations from Caragana arborescens, which grows in North America (Glawe et al. 2006; Lonergan and Skoglund 2013) and the cenopopulations in Europe (Braun 1995; Geliuta 1989; Lebeda et al. 2008; Vajna 2006). The cenopopulations of the fungus from Colutea arborescens and Robinia pseudoacacia are almost identical in terms of the range of the norm of reaction of the cleistothecium diameter (Fig. 4.). Thus, under the stressful conditions related to the invasion in new living environment, the variation range of the cleistothecium diameter decreases.
| Author | Diameter of the fungal fruit, µm | Appendages | |
| number | length, µm | ||
| Caragana arborescens Lam. | |||
| Braun (1995) | 80–140 | 5–12 | |
| Geliuta (1989) | 100–143 | Up to 16 | 152–363 |
| Glawe et al. (2006) | 100–130 | – | 145–290 |
| Lebeda et al. (2008) | 67.5–112.5 | 3–13 | 180–330 |
| Lonergan and Skoglund (2013) | 100–122 | 5–10 | – |
| Vajna (2006) | 90–160 | 9–12 | 230–312 |
| Authors | 80–145 | 6–12 | 170–295 |
| Colutea arborescens L. | |||
| Schmidt and Scholler (2002) | 86–150 | 9–12 | – |
| Robinia pseudoacacia L. | |||
| Lee and Nguyen (2018) | 85.5–122.5 | 2–8 | 113–245.5 |
| Authors | 73–123 | 6–10 | 105–280 |
Table 6. Morphometric parameters of the fungal fruit diameter and of the appendages length of the invasive species Erysiphe palczewskii on various species of nutritive plants at different ecological and geographical points.
Figure 4: The range of the norm of reaction of the fungal fruit diameter of Erysiphe palczewskii investigated by the authors on Caragana arborescens: 1–Braun 1995; 2–Geliuta 1989; 3–Glawe et al. 2006; 4–Lebeda et al. 2008; 5–Lonergan and Skoglund 2013; 6–Vajna 2006; 7–authors, on Colutea arborescens: 8–Schmidt and Scholler 2002 and on Robinia pseudoacacia: 9–Lee and Nguyen 2018; 10–Authors of the article.
The norm of reaction of the number of appendages of the fungal fruit and their length parameters of the species Erysiphe palczewskii are also not the same. Thus, the analysis of the number of appendages on one cleistothecium noted by various researchers’ shows that the fungal fruit can have from 2 (on Robinia pseudoacacia, Lee and Nguyen 2018) to 11 appendages (on Caragana arborescens, Geliuta 1989). The range of the fungal fruit of this attribute of the fungus is 9 units. However, the appendages of the fungal fruit from Colutea arborescens (Schmidt and Scholler 2002) and the fungal fruit from Caragana arborescens (Vajna 2006) showed the lowest range of number on various types of nutritive plants and at different ecological and geographical points, while the highest range of numbers of appendages (from 3 to 10) was noted on Caragana arborescens (Lebeda et al. 2008).
Analyzing the length parameters of the appendages of the fungal fruit, it should be noted that the shortest appendages (105 μm) were observed on Robinia pseudoacacia, and the longest (363 μm) on Caragana arborescens (Geliuta 1989). The range of the norm of reaction of this attribute of powdery mildew is quite broad and amounts to 258 μm. However, various species of nutritive plants at different ecological and geographical points showed different variation range of parameters of appendage length. The lowest (82 μm) was noted on Caragana arborescens (Vajna 2006), and the highest (211 μm) was also found on Caragana arborescens (Geliuta 1989).
Summarizing the data on the analysis of the variability of the fungal fruit (diameter, number and length of appendages) of Erysiphe palczewskii, it should be noted that the potential of variability of the considered attributes of the fungus is quite significant. At the same time, the indicators of the variability of the parameters of the fungal fruit diameter under stressful conditions related to the invasion in the new living environment tend to decrease. A clear dependence of influence on the indicators of the number of appendages and their length parameters from the nutritive species of plants at different ecological and geographical points was not found.
Conclusion
The investigated samples of the invasive species Erysiphe palczewskii from the host plant Caragana arborescens were characterized by a complete cycle of development. Samples of powdery mildew from Robinia pseudoacacia were characterized both by a complete cycle (conidia and cleistothecium) of the fungus development (village Glubokoe and BsZhNAEU), and by solely anamorphic cycle of development (park “Kin Grust”, Kiev).
It was established that the norm of reaction of length and width of conidia, cleistothecium diameter and the length of appendages of the invasive species Erysiphe palczewskii on various species of nutritive plants at different ecological and geographical points is not the same. The greatest differences in the range of indicators of the enumerated attributes were found between the cenopopulations of Caragana arborescens growing in North America and the cenopopulations of Europe. The cenopopulations of the fungus from Colutea arborescens and Robinia pseudoacacia were almost identical in terms of the range of the norm of reaction.
The potential of the variability of the considered signs (conidia and fungal fruit diameter) is quite considerable. At the same time, a clear dependence of influence on the indicators of the number of appendages and their length parameters from the nutritive species of plants at different ecological and geographical points was not found.
Acknowledgments
The chromatographic analysis was carried out on the equipment of the common use center “Chemistry” of UIC of the RAS.
The work was supported by the grant from the Russian Foundation for Basic Research (RFBR) 18-44-860006.
References
Braun U., Kummer V., Xu B. 2009. Taxonomy and nomenclature of powdery mildew fungi: Erysiphe asclepiadis, E. robiniicola and Golovinomyces caulicola. Mycotaxon. 107: 285-295.
Braun U. 1995. The powdery mildew (Erysephales) of Europe, Jena. NORDAC J Botany. 144-144. https://doi.org/10.1111/j.1756-1051.1996.tb00953.x
Chumak P., Palagecha R., Kovalchuk V. 2012. Morphological variability of the fungal fruit and the spread of Erysiphe magnifica (U. Braun) U. Braun and S. Takam. on the plants belonging to species Magnolia L. in the O. V. Fomin Botanical Garden. Modern Phytopathology. 2: 95-97. https://doi.org/10.5281/zenodo.162450
Geliuta V.P. 2006. Boroshnystorosiani hryby (poriadok Erysiphales) Kanivskoho pryrodnoho zapovidnyka (Erisyphaceae (order Erysiphales) of the Kaniv nature reserve). Conservation Business Ukraine. 12: 23-32.
Geliuta V.P. 1989. Fungi flora in Ukraine. Erisyphaceae. Ð?yiv (in Ukrainian).
Glawe D.A., Stack, R.W., Walla, J.A. 2006. First report of powdery mildew on Caragana arborescens in Minnesota and North Dakota caused by Microsphaera (Erysiphe) palczewskii. Plant Health Progress. 7. http://dx.doi.org/10.1094/PHP-2006-0117-01-BR
Huhtinen S., Alanko P., Makinen Y. 2001. The invasion history of Microsphaera palczewskii (Erysiphales) in Finland. Karstenia. 4: 31-36. http://www.funga.fi/Karstenia/Karstenia%2041-1%202001-5.pdf
Jones D.R., Baker R.H.A. 2007. Introductions of non-native plant pathogens into Great Britain, 1970-2004. Plant Pathology. 56: 891-910. https://doi.org/10.1111/j.1365-3059.2007.01619.x
Lebeda A., Mieslerová B., SedláÅ?ová M. et al. 2008. Occurrence of anamorphic and teleomorphic stage of Erysiphe palczewskii (syn. Microsphaera palczewskii) on Caragana arborescens in the Czech Republic and Austria and its morphological characterisation. Plant Protect Sci. 44: 41-48. https://www.agriculturejournals.cz/publicFiles/20_2008-PPS.pdf
Lee H.B., Nguyen T.T. 2019. First report of powdery mildew caused by Erysiphe palczewskii on Robinia pseudoacacia in Korea. Plant Disease. 103: 1428. https://doi.org/10.1094/PDIS-12-18-2144-PDN
Lonergan E., Skoglund L.G. 2013. First report of powdery mildew (Microsphaera palczewskii) on Siberian peashrub (Caragana arborescens) in Montana. Plant Health Progress. 14. https://doi.org/10.1094/PHP-2013-0327-01-BR
Nischwitz C., Newcombe G. 2003. First report of powdery mildew (Microsphaera palczewskii) on Siberian pea tree (Caragana arborescens) in North America. Plant Disease. 87: 451. https://doi.org/10.1094/PDIS.2003.87.4.451B
Palagecha P.M., Chumak P.Ya. 2011. Ecological and morphological structure of subpopulation Microsphaera magnifica U. Braun (Erysiphales) and invasive capability of this fungus. Proceedings of ХIII convention of Ukrainian Botanic Society. pp: 313.
Shevchenko S.V., Tsyliurik A.V. 1986. Forest phytopathology. Kiev (in Ukrainian).
Smidt A., Scholler M. 2002. Studies in Erysiphales anamorphs (II): Colutea arborescens, a new host for Erysiphe palczewskii. Feddes Repertorium. 113: 107-111. https://doi.org/10.1002/1522-239X(200205)113:1/2%3C107::AID-FEDR107%3E3.0.CO;2-1
Takamatsu S., Ito H., Shiroga Y. et al. 2015. First comprehensive phylogenetic analysis of the genus Erysiphe (Erysiphales, Erysiphaceae) II: The Uncinula lineage. Mycologia. 107: 475-489. https://doi.org/10.3852/15-007
Vajna L. 2006. First report of powder mildew on Caragana arborescens in Hungary caused by Erysiphe palczewskii. New Disease Reports. 3: 14. https://www.ndrs.org.uk/article.php?id=013014