Research - Modern Phytomorphology ( 2020) Volume 14, Issue 2
Features of leaf mesostructure organization under plant growth regulators treatment on broad bean plants
O. A. Shevchuk1*, O. O. Kravets1, V. V. Shevchuk2, O. O. Khodanitska1, O. O. Tkachuk1, L. A. Golunova1, S. V. Polyvanyi1, O. V. Knyazyuk1 and O. L. Zavalnyuk12Vinnytsia National Agrarian University, Soniachna Str. 3, Vinnytsia, 21008, Ukraine
O. A. Shevchuk, Vinnytsia Mykhailo Kotsiubynskyi State Pedagogical University, Ostrozhskogo Str. 32, Vinnytsia, 21000, Ukraine, Email: shevchukoksana8@gmail.com
Received: 04-Mar-2020 Accepted: 18-Jul-2020 Published: 27-Jul-2020
Abstract
It was studied the mesostructure organization of leaf apparatus and pigment content of leaves under application of growth regulators with a different mechanism of action-chlormequat chloride (1%) (retardant of quaternary ammonium compounds group) and Emistim C (0,1 ml/l) (growth stimulator) on broad bean plants. The plant growth regulators treatment on broad beans led to the thickening of leaves due to an increase in the growth of columnar and spongy leaf parenchyma. Growth regulators influenced the formation of the stomatal apparatus of broad beans leaves in different ways. The inhibitory compound chlormequat chloride caused a decrease in the number of lower epidermal cells and stomata with a simultaneous increase in the area of the stomatal cell. The application of stimulator compound Emistim C led to an increase in epidermal cells, without differences in the number of stomata but the area of stomatal cells increased significantly. It has been established the enhancement of photosynthetic processes due to an increase in the chlorophyll content in assimilative cells of leaves under chlormequat chloride on broad beans.
Keywords
Plant growth regulators, donor-acceptor system (“source-sink”), mesostructure organization, stomatal apparatus, chlorophyll, broad beans
Introduction
The plant organism can be represented as the only donor-acceptor system (“source-sink”), in which the plant is considered as a system of donors (source) and acceptors (sink) of assimilates. Donors and acceptors can be considered as individual plant structuresorgans, tissues, cells and organelles, and as processesphotosynthesis, respiration, transport, assimilation (Kiriziy et al. 2014). It is known that regulation of the source-sink system of a plant organism can be carried out artificially by growth regulators application, it causes an increase in the crop production process (Kuryata and Khodanitska 2018; Shevchuk et al. 2019) and improves crop quality (Khodanitska et al. 2019) due to redistribution of assimilating flow to economically valuable organs. Biologically active substances act on the regulatory mechanisms in cells of a plant organism at the metabolic level (Bhatla 2018).
One of the modern methods to increase the crop yield is an application of synthetic plant growth regulators that are made on a natural substance basis and act similarly to phytohormones (Kuryata et al. 2019; El Karamany 2019; Khodanitska et al. 2019). These compounds are environmentally friendly and positive effects on the microflora of soil, increase the resistance of plants to adverse environmental factors (extreme temperatures, drought and heat resistance, and frost resistance) (Khan et al. 2019). The most common groups of synthetic plant growth regulators are retardants–antigibberellin compound. Retardants are widely used to increase the yield of oilseeds (Kuryata and Khodanitska 2018; Khodanitska et al. 2019; Kuryata et al. 2019), vegetables (Kuryata and Kravets 2018), legumes (Didur et al. 2019; Kuryata et al. 2019; Mazur et al. 2019) and industrial crops (Shevchuk et al. 2019).
It is necessary to make attention to legumes due to the growing deficiency of protein. The value of these crops lies in the fact that they not only increase the resources of food and feed grains but also increase soil fertility and crop production (Pankievicz et al. 2019). The broad bean plants have a great importance among legumes grown in our country for the grains and green manure. They produce high yields on heavy clay soils compared to peas and fodder lupins, fix nitrogen from the atmosphere and are a good predecessor for winter and spring wheat. In this case, the issue of this study was to establish the application of stimulator Emistim C and inhibitor chlormequat chloride on the mesostructure features of the leaf apparatus of broad bean plants.
Materials and Methods
Plant material
Broad bean plants cv. Vizir was treated with aqueous solutions of chlormequat chloride (∇∇∇-720) (1%) and growth stimulator Emistim C (0,1 ml/l) to complete the wetting of leaves at shoot height 10 cm-15 cm. Mesostructure organization of leaves was studied on a fixed material according to the generally accepted method of Mokronosov A.T. and Borzenkova R.A.
Data analysis
It was used a mixture of equal parts of ethanol, glycerol and water with the addition of 1% formalin for the preservation of leaves. The maceration agent was a 5% solution of acetic acid in 2 mol/l hydrochloric acid. The method of partial maceration of leaf tissue was used to determine epidermal cells. Measurement of the area of epidermal cells was carried out by using a microscope and an ocular micrometer MOB-1-15x. The number of cells in the tissue per unit area of the field of view was calculated with a followed calculation of one cell and its volume. The chlorophyll content was determined in the fresh matter by spectrophotometric method on the spectrophotometer SF-16. The analytical repeatability of the research is fivefold (AOAS, 2010). The statistical processing of results was performed by the method of one-way analysis of variance.
Results
Features of mesostructure organization of leaves
The results of the study of the anatomical structure of leaves indicate the restructuring of leaf apparatus of treated plants under application of growth inhibitor chlormequat chloride and stimulator Emistim C on broad beans (Tab. 1.). It was observed that the thickness of the leaf blade of treated plants increased due to the growth of parenchyma. It was found that both growth regulators increased the volume of columnar cells and the size (length and width) of spongy parenchyma cells of leaves.
| Measurements | Control | Chlormequat chloride | Emistim С |
| Thickness of leave, μm | 231,6 ± 0,17 | 250,4 ± 2,35* | 260,1 ± 8,48* |
| Volume of palisade parenchyma, μm3 | 3997,6 ± 72,03 | 4645,6 ± 62,61* | 6387,4 ± 64,63* |
| Length of spongy cells, μm | 25,9 ± 1,18 | 28,7 ± 1,12 | 28,2 ± 0,62 |
| Width of spongy cells, μm | 17,9 ± 0,72 | 18,3 ± 0,46 | 19,1 ± 0,78 |
| Number of stomata on 1 mm2 of the abaxial leaf surface, pieces | 277,6 ± 10,95 | 193,6 ± 5,60* | 287,8 ± 6,11 |
| Area of a stomata, μm2 | 421,6 ± 7,24 | 450,0 ± 8,87* | 472,7 ± 6,92* |
| Number of epidermal cells on 1 mm2 of the abaxial leaf surface, pieces | 705,5 ± 9,42 | 468,6 ± 10,69* | 817,5 ± 10,44 |
Note: *difference is significant at p<0,05
Table 1. Leaf mesostructure indicators under plant growth regulators treatment on the broad bean.
It was found an increase of columnar parenchyma that is the main assimilation tissue of leaves. The stomata of the lower epidermis are important for the transpiration processes–a physiological process that creates a continuous flow of water from the root system to leaves, combining all plants in a single system, and protects the plant organism from overheating.
Plant growth regulators influenced the formation of the stomatal apparatus of broad bean leaves. The number of stomata decreased under chlormequat chloride treatment and their number remained practically unchanged under Emistim C. The application of inhibitory compound chlormequat chloride led to a decrease in the epidermal cells and the number of stomata with a simultaneous increase in the stomatal cell area. The treatment of stimulator Emistim C led to an increase in the number of epidermal cells, but the number of stomata changed and the area of stomatal cells increased significantly.
Leaf pigment content
The effectiveness of photosynthesis is determined by the functional state of the photosynthetic apparatus, one of the indicators of which is the pigment content. An increase in photosynthetic processes at the beginning of vegetation can be indicated by an increase in the chlorophyll content of broad bean plants (Fig. 1.). Application of chlormequat chloride increased in the pigment content by 29% compared to control. Growth stimulator Emistim C not affected on the pigment content, it was close to control at the last stages of vegetation.
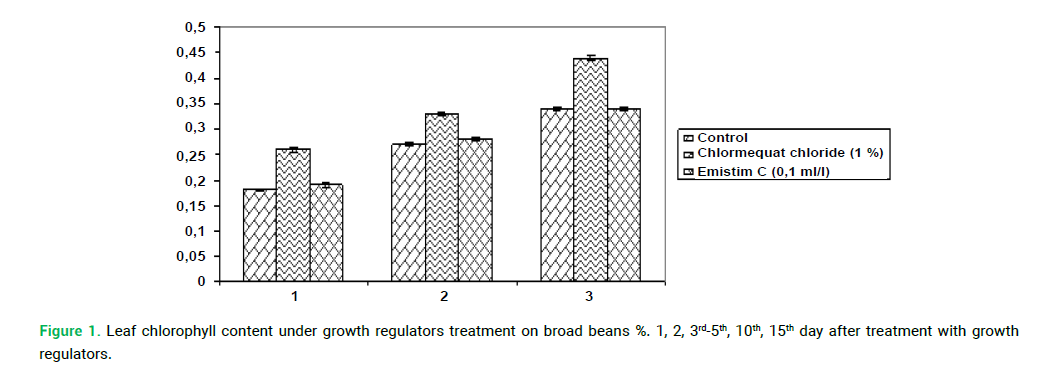
Figure 1: Leaf chlorophyll content under growth regulators treatment on broad beans %. 1, 2, 3rd-5th, 10th, 15th day after treatment with growth regulators.
Discussion
The analysis of mesostructural indicators of photosynthetic apparatus of broad bean plants confirmed that chlormequat chloride and growth stimulator Emistim C enhanced the formation of photosynthetic apparatus the number of stomata increased the area of a stoma and the thickness of leaves due to the growth of chlorenchyma. It was noted that chlormequat chloride treatment increased in photosynthetic processes due to the chlorophyll accumulation in assimilative cells of broad bean leaves. It is advisable to increase the crop production of broad bean plants by chlormequat chloride
Conclusion
Application of plant growth regulators is a powerful method to regulate the activity of assimilative apparatus, as one of the main components of donor-acceptor system of plants, and can be used for targeted regulation of plastic substances redistribution in board beans, that increased in the number of stomata, the area of a stoma, and the thickness of leaves due to the growth of chlorenchyma. It testifies to the fact that chlormequat chloride treatment on broad bean plants leads to an increase in the leaf chlorophyll content and improve photosynthetic processes.
References
AOAC. 2010. Official methods of analysis of association of analytical chemist international 18th ed. Rev. 3. 2010. Ass of Analytical Chemist. Gaithersburg, Maryland, USA. https://www.worldcat.org/title/official-methods-of-analysis-of-aoac-international/oclc/62751475
Bhatla S.C. 2018. Plant growth regulators: an overview. In: plant physiology, development and metabolism. Springer, 559-568. https://doi.org/10.1007/978-981-13-2023-1_1
Didur, I.M., Prokopchuk, V.M., Pantsyreva H.V. 2019. Investigation of biomorphological and decorative characteristics of ornamental species of the genus Lupinus L. Ukr J Ecol 9: 287-290. https://doi.org/10.15421/2019_743
El Karamany M.F., Sadak M.S., Bakry B.A. 2019. Improving quality and quantity of mungbean plant via foliar application of plant growth regulators in sandy soil conditions. Bull Natl Res Cent 43. https://doi.org/ 10.1186/s42269-019-0099-5
Kiriziy D.A., Stasyk О.О., Pryadkina G.А., Shadchyna Т.М. 2014. Fotosintez. Assimilyatsiya СО2 i mehanizmy jejyo regulyatsii. Kiev.
Khan N., Bano A., Babar, M.A. 2019. The stimulatory effects of plant growth promoting rhizobacteria and plant growth regulators on wheat physiology grown in sandy soil. Arch Microbiol 201: 769-785. https://doi.org/10.1007/s00203-019-01644-w
Khodanitska O.O., Kuryata V.G., Shevchuk O.A. Tkachuk O.O., Poprotska I.V. 2019. Effect of treptolem on morphogenesis and productivity of linseed plants. Ukr J Ecol 9: 119-126. https://www.ujecology.com/articles/effect-of-treptolem-on-morphogenesis-and-productivity-of-linseed-plants.pdf
Khodanitska О.О., Shevchuk O.A., Tkachuk O.O., Shevchuk V.V. 2019. Features of the anatomical structure of the autonomic organs and flax oil yield (Linum usitatissimum L.) at applications growth stimulants. Sci Rise: Biol Sci 4: 35-40. https://doi.org/10.15587/2519-8025.2019.188317
Kuryata V.G., Golunova L.A., Poprotska I.V., Khodanitska O.O. 2019. Symbiotic nitrogen fixation of soybean-rhizobium complexes and productivity of soybean culture as affected by the retardant chlormequat chloride. Ukr J Ecol 9: 5-13. http://socrates.vsau.org/repository/getfile.php/23050.pdf
Kuryata V.G., Khodanitska O.O. 2018. Features of anatomical structure, formation and functioning of leaf apparatus and productivity of linseed under chlormequat chloride treatment. Ukr J Ecol 8: 918-926. https://doi.org/10.15421/2018_294
Kuryata V.G., Kravets O.O. 2018. Features of morphogenesis, accumulation and redistribution of assimilate and nitrogen containing compounds in tomatoes under retardants treatment. Ukr J Ecol 8: 356-362. https://doi.org/10.15421/2018_222
Kuryata V.G., Polyvanyi S.V., Shevchuk O.A. 2019. Morphogenesis and the effectiveness of the production process of oil poppy under the complex action of retardant chlormequat chloride and growth stimulant treptolem. Ukr J Ecol 9: 127-134.
Mazur V.A., Pantsyreva H.V., Mazur K.V., Didur I.M. 2019. Influence of the assimilation apparatus and productivity of white lupine plants. Agron Res 17: 206-219. https://doi.org/10.15159/AR.19.024
Pankievicz V.C.S., Irving T.B., Maia L.G.S. 2019. Are we there yet? The long walk towards the development of efficient symbiotic associations between nitrogen-fixing bacteria and non-leguminous crops. BMC Biol 17: 99. https://doi.org/10.1186/s12915-019-0710-0
Shevchuk O.A., Tkachuk O.O., Kuryata V.G., Khodanitska O.O., Polyvanyi S.V. 2019. Features of leaf photosynthetic apparatus of sugar beet under retardants treatment. Ukr J Ecol 9: 115-120. https://www.ujecology.com/articles/features-of-leaf-photosynthetic-apparatus-of-sugar-beet-under-retardants-treatment.pdf