Research Article - Modern Phytomorphology ( 2026) Volume 20, Issue 2
Effects of biochar on the root morphological and mechanical properties of Lespedeza bicolor Turcz. on slopes in high-altitude areas
Yue Kong1, Xiao Tan2, Junzhuo Li2, Yuanyuan Song1, Jiayi Yang1, Qingyue Liu1 and Hailong Sun1*2Sichuan Highway Planning, Survey, Design and Research Institute Ltd., Chengdu 610065, China
Hailong Sun, State Key Laboratory of Hydraulics and Mountain River Engineering, College of Water Resources and H, Sichuan University, Chengdu 610065, China, Email: sunhl@scu.edu.cn
Received: 23-Mar-2026, Manuscript No. mp-26-186737; Accepted: 16-Apr-2026, Pre QC No. mp-26-186737 (PQ); Editor assigned: 25-Mar-2026, Pre QC No. mp-26-186737 (PQ); Reviewed: 08-Apr-2026, QC No. mp-26-186737; Revised: 15-Apr-2026, Manuscript No. mp-26-186737 (R); Published: 23-Apr-2026, DOI: 10.5281/zenodo.19689284
Abstract
To investigate the effects of biochar on the root growth and soil reinforcement capacity of slope vegetation in high-altitude regions, in this study, Yajiang County, Garze Tibetan Autonomous Prefecture, Sichuan Province, was selected as the experimental area, and <i>Lespedeza bicolor</i> was selected as the research object. Six biochar application gradients were established: 0 kg·m-2 (CK), 2.5 kg·m-2 (I), 5 kg·m-2 (II), 7.5 kg·m-2 (III), 10 kg·m-2 (IV), and 12.5 kg·m-2 (V). The effects of different application rates on the soil physical and chemical properties, root morphology, chemical composition, and tensile strength of L. bicolor were systematically analyzed. The results indicated that the biochar application rate significantly affected the root morphological and mechanical properties. Under Treatment III (7.5 kg·m-2), the root length, root biomass, total root length, and specific root length of <i>L. bicolor</i> were significantly greater than those under the other treatments. Moreover, this treatment resulted in the highest tensile strength of the secondary lateral roots, which was accompanied by an increase in cellulose content and a decrease in lignin content. Root tensile strength was positively correlated with cellulose content and negatively correlated with lignin content. In conclusion, an appropriate amount of biochar can promote root development and increase its tensile strength by improving soil conditions and regulating root chemical composition. Among the tested concentrations, 7.5 kg·m-2 was the optimal biochar application rate for enhancing the soil reinforcement capacity of the roots of <i>L. bicolor</i> in this region. This study provides theoretical support and a practical reference for slope ecological restoration in high-altitude areas.
Keywords
Biochar, High-altitude slopes, Root growth, Mechanical properties
Introduction
In recent years, the continuous expansion of transportation, energy, and other infrastructure projects into high-altitude areas has boosted socioeconomic development, yet has also exerted certain adverse impacts on the ecological environment. High-altitude regions are characterized by infertile soil, low soil fertility, and fragile ecosystems. As a result, disturbances caused by engineering activities tend to induce severe soil erosion and slope exposure, which not only undermine the stability of the ecosystem but also pose potential risks to the safety and durability of infrastructure (Sun, et al. 2023). Accordingly, slope restoration has become an urgent issue to be addressed.
Vegetation restoration is an important measure for slope ecological restoration. The presence of roots not only increases soil particle friction and cohesion (Dupuy, et al. 2005), thereby enhancing the anchoring effect of plant roots, but also mitigates soil erosion induced by water flow (De Baets and Poesen 2010). Consequently, these root-induced effects improve the shear resistance of slopes and minimize deformation caused by slope instability (Eab, et al. 2015). However, the full exertion of these mechanical effects by roots largely depends on the physical structure and fertility of the slope soil. The particularity of high-altitude regions severely restricts the effect of vegetation restoration (Wang, et al. 2024). High-altitude areas in China exhibit distinct characteristics including drastic fluctuations in hydrothermal conditions, inferior soil quality, and slow nutrient cycling. These factors greatly limit vegetation growth and can hardly meet the demands of slope vegetation restoration (Barberis, et al. 2023). Therefore, the development of efficient restoration materials capable of improving soil properties in high-altitude areas and promoting plant root growth has become a breakthrough to enhance slope remediation.
Biochar is a cost-effective soil amendment that has been widely used for the management and restoration of degraded soils (El-Naggar, et al. 2018). Owing to its unique porous structure and physicochemical properties, biochar not only enhances soil nutrient retention and water-holding capacity but also influences plant root morphology and mechanical characteristics (Coomes and Miltner 2017, Xiang et al. 2017). Numerous studies have so far focused on the application of biochar in soils and low-altitude agricultural systems (Oram, et al. 2014, Isimikalu, et al. 2023, Murtaza, et al. 2023), and the results have confirmed its significant effects on improving crop yield and ameliorating degraded soils (Major, et al. 2010, Sadaf, et al. 2017, Murtaza, et al. 2021, Bezzalla, et al. 2026). In recent years, biochar has been widely used in slope ecological restoration due to its significant advantages in improving soil structure and enhancing soil nutrient and water retention capacities, which provides a new strategy for strengthening the soil-fixing ability of slope vegetation (Sharma, 2024). Nevertheless, research on the mechanical properties of plant roots under engineering disturbance in high-altitude regions remains relatively limited. Particularly in ecologically fragile alpine areas, there is still a lack of empirical studies to evaluate the regulatory mechanism of biochar on the mechanical properties of plant roots and its relationship with the synergistic effects of soil water and fertilizer. Therefore, exploring the effects of biochar on the mechanical properties of plant roots can not only provide a theoretical basis for the synergistic regulation of water and fertilizer in high-altitude regions, but also be of great practical significance for slope vegetation restoration.
The Yalong River Basin belongs to the plateau alpine gorge region (Li, et al. 2025). Its fragile ecosystem and infertile soil make vegetation restoration extremely difficult. Lespedeza bicolor Turcz. is an adaptable leguminous shrub. Aiming to address the difficulty of natural vegetation restoration in the Yalong river basin, six treatments with different biochar application rates were set up in Yajiang county, Garze Tibetan autonomous prefecture, Sichuan province. We systematically determine and analyze the morphological traits, chemical constituents, and mechanical properties of L. bicolor roots. The following hypotheses were proposed: (1) Biochar application would significantly affect the root morphological and mechanical properties of L. bicolor; (2) There is a threshold for biochar application rate. Excessive biochar may destroy the soil physicochemical properties, disturb the balance between soil nutrient supply and plant uptake, and thus inhibit plant growth.
Materials and Methods
Study site
The study site is located in Yajiang County, Garze Tibetan autonomous prefecture, Sichuan province, southwestern China. It lies in the middle reaches of the Yalong river basin, within the Hengduan Mountains region of the Qinghai-Tibet Plateau. Yajiang County belongs to the subhumid climate zone of the Qinghai-Tibet Plateau, characterized by a continental monsoon alpine climate. The annual average temperature is 11.1°C, with monthly average temperatures ranging from 1.4°C to 18.0°C, and the annual average sunshine duration reaches 2319 hours-characterized by abundant sunshine, intense solar radiation, and a significant diurnal temperature variation. The annual average precipitation is 783.1 mm, and the annual average relative humidity is 54.4%; the winter and spring seasons are cold and arid, with no distinct summer period. Hydropower development in the Yalong river basin has resulted in extensive bare slopes, which are highly susceptible to geological hazards such as landslides. Consequently, ecological restoration of these slopes is of urgent necessity. In this study, one representative slope was selected as the research object, and its location and fundamental parameters are illustrated in Fig. 1.
Figure 1: Effect of different biochar application rates on plant growth status. (a) Plant height; (b) Basal diameter; (c) Number per unit area. Data are presented as the means ± standard deviations. Control (CK), I, II, III, IV, and V represent biochar application rates of 0, 2.5, 5, 7.5, 10, and 12.5 kg·m-2, respectively. Lowercase letters indicate statistically significant differences among different biochar content treatments at the 0.05 significance level.
Experimental design
Experimental materials: The biochar material was provided by Liao Ning Golden Future Agriculture Technology Co., Ltd. It was produced via high-temperature carbonization of rice straw at 500°C under anaerobic conditions. The basic physicochemical properties of the biochar were as follows: Specific surface area of 8.85 m2·g-1, average pore diameter of 16.25 nm, pH 9.32, ash content of 12.6%, total nitrogen content of 0.6%, available phosphorus content of 20.89 g·kg-1, and available potassium content of 2.51 g·kg-1.
Plant materials: Lespedeza bicolor, provided by Yunnan Jinye Ecological Construction Group Co., Ltd.
Experimental setup: Given the shallow topsoil on the experimental slope, slope vegetation restoration was carried out using the foreign soil covering method, with a covering thickness of 12 cm (the basic physicochemical properties of the foreign soil are presented in Tab. 1). The foreign soil was mixed with L. bicolor seeds at a sowing density of 15 g·m-2. The experiment was established in early May 2016, with conventional water management applied during the entire test period. Upon completion of the experiment in May 2024, plant growth conditions were investigated and the relevant indicators were measured.
| Parameter | |
|---|---|
| pH | 6.17 ± 0.06 |
| Organic matter (g·kg-1) | 18.67 ± 4.12 |
| Available nitrogen (mg·kg-1) | 44.23 ± 7.28 |
| Available phosphorus (mg·kg-1) | 64.21 ± 6.19 |
| Available potassium (mg·kg-1) | 118.66 ± 8.36 |
| Note: Data are presented as the means ± standard deviations. | |
Table 1. Basic physical and chemical properties of foreign soil (when biochar is not added).
A Randomized Complete Block Design (RCBD) was adopted, comprising six biochar application treatments: Control (CK, 0 kg·m-2), Treatment I (I, 2.5 kg·m-2), Treatment II (II, 5 kg·m-2), Treatment III (III, 7.5 kg·m-2), Treatment IV (IV, 10 kg·m-2), and Treatment V (V, 12.5 kg·m-2). Each treatment was assigned to a 5 m × 5 m plot, with three replicates per treatment, resulting in a total of 18 standard plots. To avoid mutual interference, a spacing of 3 m was maintained between adjacent plots.
Measurement and calculation of indicators
Measurement of plant growth status: The number of L. bicolor individuals in each quadrat was counted. Three plants were randomly selected from each quadrat to measure plant height and basal diameter. Plant height was measured as the straight-line distance from the rhizome node to the plant apex using a measuring tape with an accuracy of 1 mm. The mean value of repeated measurements was taken as the plant height for each individual. Basal diameter was determined at the soil surface using a Vernier caliper with an accuracy of 0.01 mm. Measurements were conducted in two mutually perpendicular directions, and the average of the two values was recorded as the basal diameter for each individual. Data on plant height and basal diameter of the selected individual plants within each treatment group were statistically analyzed separately. The mean values within each group were calculated and used as the representative values of plant height and basal diameter of L. bicolor under different biochar application treatments.
Measurement of root morphology: After selecting the L. bicolor individuals for excavation, surface debris including fallen leaves, gravel, and loose topsoil around the plant base was carefully removed. The position where the rhizome contacts the soil surface was marked, and the aboveground part of the plant was moderately pruned to lower its center of gravity and reduce tensile damage to the root system caused by external forces during excavation. During excavation, soil removal started from the surface layer and proceeded layer by layer from top to bottom and from outside to inside. Gentle digging and stripping were adopted to avoid root breakage caused by forced pulling. Upon completion of excavation, the root systems were observed. All plants exhibited well-developed root systems with distinct taproots, primary lateral roots, and secondary lateral roots. Subsequently, the number, length (distance from the growth point to the root tip), and average diameter of first-order and second-order lateral roots for each plant were determined. The average diameter was measured with a vernier caliper at 10 cm from the root attachment point, as the mean of the maximum and minimum diameters in two perpendicular directions. The length and diameter of the taproots were also measured, and fibrous roots on the lateral roots were collected. Based on the measured data, the mean values of taproot length and diameter, as well as the mean values of the number, length, and diameter of first-order lateral roots and second-order lateral roots for each individual plant were calculated. After root excavation and measurement, impurities adhering to the root surfaces were carefully removed. The different root types were classified, packaged, and stored in a refrigerator at 4°C for low-temperature preservation, and all subsequent tests were completed within 7 days.
Measurement of root tensile strength: For each plant root sample, the tensile strength of the taproot, first-order lateral roots, and second-order lateral roots was determined separately. Three replicates were randomly selected for both first-order lateral roots and second-order lateral roots. All test root segments were prepared according to a uniform standard: the segment length was fixed at 10 cm, with only one segment taken per root; segments were cut starting at 5 cm from the root growth point. Prepared root segments were immediately subjected to tensile resistance testing using an electronic universal testing machine (KD III, Shenzhen Kaiqiangli Testing Instrument Co., Ltd.).
Root tensile strength was calculated from the measured data, using the following formula:
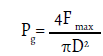
Where Pg is the tensile strength of the root (MPa), Fmax is the maximum tensile force (N), and D is the diameter of the root segment (mm).
Determination of root biomass and chemical constituents: The plant roots (including those used for tensile strength testing) were oven-dried at 80°C for 48 h and then weighed to determine the root dry weight per plant. The contents of cellulose, hemicellulose, and lignin in the taproots, first-order lateral roots, and second-order lateral roots were determined via the nitric acid-ethanol method (Chen, et al. 2018), the hydrochloric acid hydrolysis-DNS method (Xiong, et al. 2005), and the 72% concentrated sulfuric acid method (Zhang, et al. 2014), respectively.
Collection and analysis of soil samples: Soil samples were collected using a cutting ring. During sampling, impurities such as stones, crop roots, and debris were removed from the samples. After sampling, the samples were sealed immediately and transported to the laboratory for analysis. The soil samples were air-dried naturally, ground, and then passed through a 2 mm nylon sieve. Soil saturated water content and field capacity were measured via the cutting ring method (Ma, et al. 2024). The soil pH value was measured by the potentiometric method with a soil-to-water ratio of 1:2.5; available nitrogen was determined via the alkali-hydrolysis diffusion method (Li, et al. 2021); available phosphorus was determined via the 0.5 mol.L-1 NaHCO3 extraction-molybdenum antimony anticolorimetric method (Scrimgeour, 2008); and available potassium was measured via the NH4OAc extraction-flame photometry method (Zanati, et al. 1973).
Statistical analysis
Data analysis was performed using SPSS 26.0. According to different sample sizes, the Kolmogorov-Smirnov test was used to test the normality of plant root traits (n>50), and the Shapiro-Wilk test was used to test the normality of soil properties (n<50). The homogeneity of variance was assessed via the Levene test. One-way Analysis Of Variance (ANOVA) was conducted for data satisfying normality and homogeneity of variance. The Tukey test was used for post hoc multiple comparisons among different biochar application treatments, and the LSD test was used for post hoc multiple comparisons among different root types. For data that violated the homogeneity of variance assumption, the Welch test was applied, followed by the Games-Howell test for post hoc multiple comparisons. The graphs were generated using Origin 2024.
Results
Plant growth status
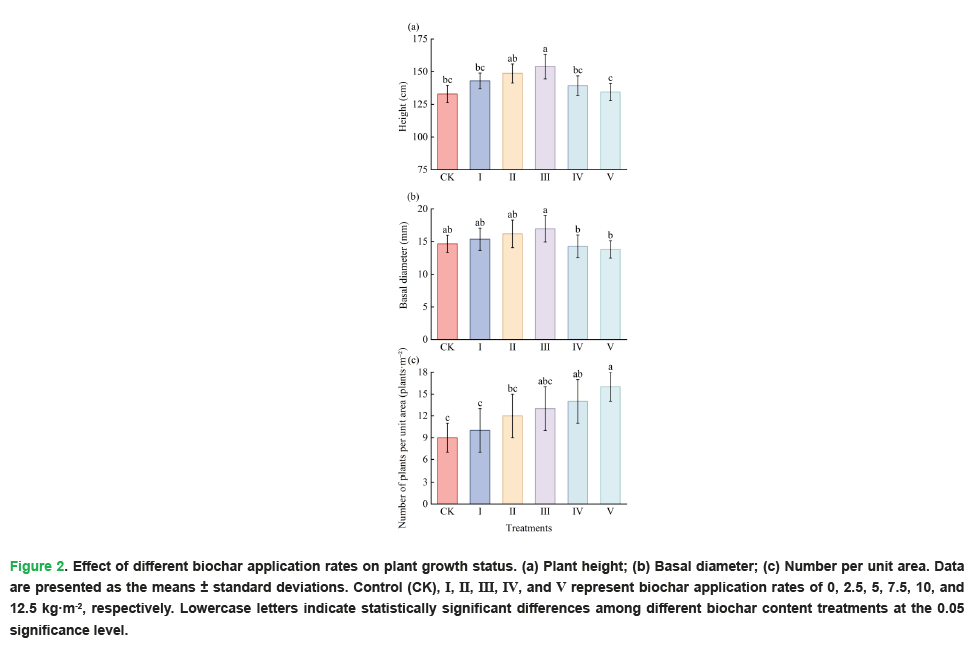
As shown in Fig. 2, plant height (P=0.000) and basal diameter (P=0.002) increased first and then decreased with increasing biochar application rate, reaching the maximum under Treatment III. Moreover, Treatment III was significantly higher than Treatments IV and V. In addition, plant height under Treatment III was significantly higher than that under Control (CK) and Treatment I, while there were no significant differences in basal diameter among Control (CK), Treatments I, II and III. The quantity per unit area increased with increasing biochar application rate (P=0.000). Compared with Control (CK), no significant increases were observed in Treatments I, II and III, while significant differences were found in Treatments IV and V.
Figure 2: Effect of different biochar application rates on plant growth status. (a) Plant height; (b) Basal diameter; (c) Number per unit area. Data are presented as the means ± standard deviations. Control (CK), I, II, III, IV, and V represent biochar application rates of 0, 2.5, 5, 7.5, 10, and 12.5 kg·m-2, respectively. Lowercase letters indicate statistically significant differences among different biochar content treatments at the 0.05 significance level.
Soil physical and chemical properties
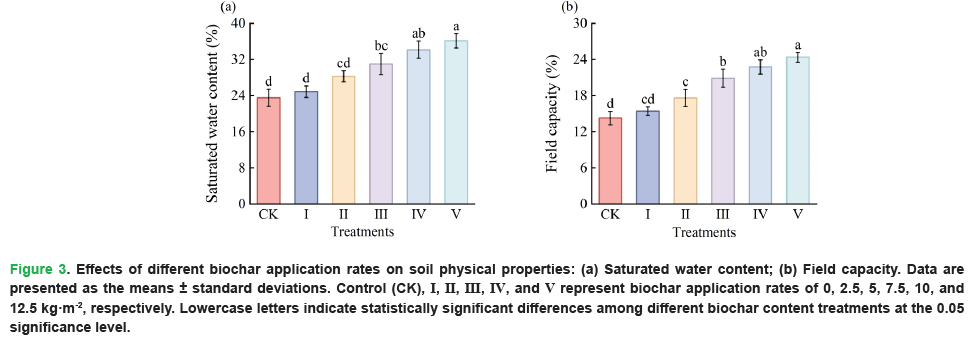
Different biochar application treatments caused significant differences in soil saturated water content (P=0.000) and field capacity (P=0.000). As shown in Fig. 3, compared with the control, Treatments I to V increased soil saturated water content and field capacity during the growth period of L. bicolor to varying degrees, with an overall increasing trend.
Figure 3: Effects of different biochar application rates on soil physical properties: (a) Saturated water content; (b) Field capacity. Data are presented as the means ± standard deviations. Control (CK), I, II, III, IV, and V represent biochar application rates of 0, 2.5, 5, 7.5, 10, and 12.5 kg·m-2, respectively. Lowercase letters indicate statistically significant differences among different biochar content treatments at the 0.05 significance level.
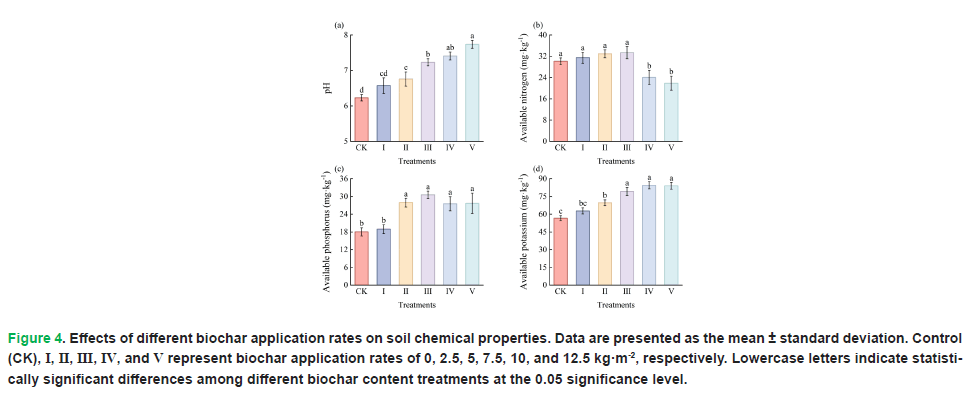
Different biochar application treatments also resulted in significant differences in soil available nitrogen (P=0.000), available phosphorus (P=0.000), available potassium (P=0.000), and soil pH (P=0.000). Fig. 4 shows that soil pH increased significantly and continuously with increasing biochar application rate, indicating that biochar addition significantly increased soil pH. Available nitrogen exhibited a trend of increasing first and then decreasing with increasing biochar application rate, reaching the maximum under Treatment III. However, compared with Control (CK), available nitrogen contents in Treatments I, II and III showed no significant increase, while those in Treatments IV and V decreased significantly. Available phosphorus increased significantly with increasing biochar application rate, peaking under Treatment III. Compared with Control (CK), no significant increase was observed in Treatment I, while significant increases were found in Treatments II, III, IV and V, but there were no significant differences between Treatment III and Treatments II, IV and V. Biochar application significantly increased available potassium content. Compared with CK, significant differences were detected in Treatments II, III, IV and V, but no significant differences were observed between Treatment III and Treatments IV and V.
Figure 4: Effects of different biochar application rates on soil chemical properties. Data are presented as the mean ± standard deviation. Control (CK), I, II, III, IV, and V represent biochar application rates of 0, 2.5, 5, 7.5, 10, and 12.5 kg·m-2, respectively. Lowercase letters indicate statistically significant differences among different biochar content treatments at the 0.05 significance level.
Root morphological characteristics
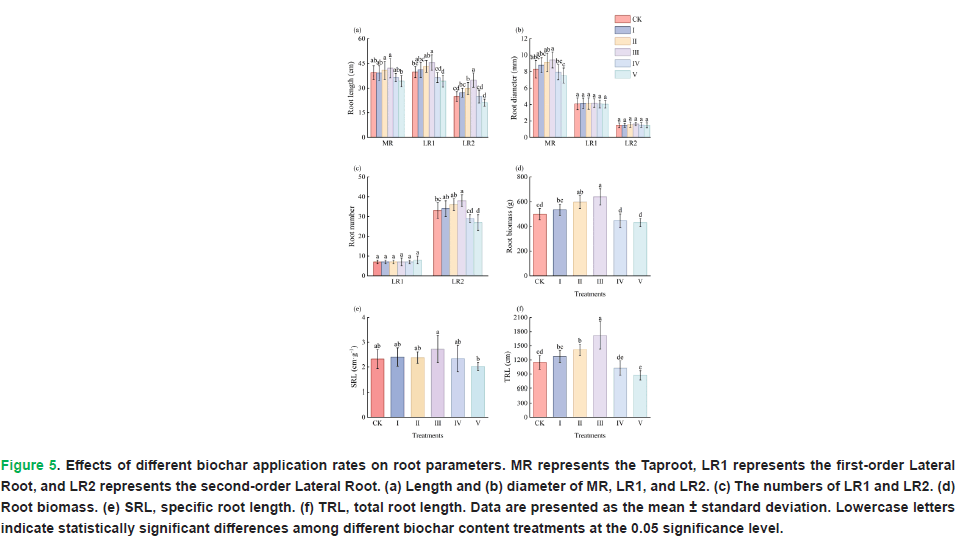
According to the ANOVA results, among the root morphological traits investigated, different biochar application rates significantly affected Taproot Length (TRL) (P=0.005) and Taproot Diameter (TRD) (P=0.001). Significant differences were observed in the length of first-order lateral roots (P=0.000), as well as in the length (P=0.000) and number (P = 0.000) of second-order lateral roots across the different biochar application rates. In contrast, the diameter (P=0.996) and number (P=0.990) of first-order lateral roots, as well as the diameter of second-order lateral roots (P=0.807), were not significantly affected by the biochar application rate. Taproot length, taproot diameter, first-order lateral root length, and second-order lateral root length and number were significantly greater in Treatments II and III than in Treatments IV and V.
Different biochar application rates significantly affected the root biomass (P=0.001), total root length (P=0.001), and specific root length (P=0.025) of L. bicolor. The root biomass in Treatments II and III was significantly greater than that in Treatments IV and V. The total root length in Treatment â ¢ were significantly greater than those in the other treatments. Additionally, the SRL values were similar among the Control (CK) and Treatments I, II , and IV (Fig. 5).
Figure 5: Effects of different biochar application rates on root parameters. MR represents the Taproot, LR1 represents the first-order Lateral Root, and LR2 represents the second-order Lateral Root. (a) Length and (b) diameter of MR, LR1, and LR2. (c) The numbers of LR1 and LR2. (d) Root biomass. (e) SRL, specific root length. (f) TRL, total root length. Data are presented as the mean ± standard deviation. Lowercase letters indicate statistically significant differences among different biochar content treatments at the 0.05 significance level.
Root chemical composition
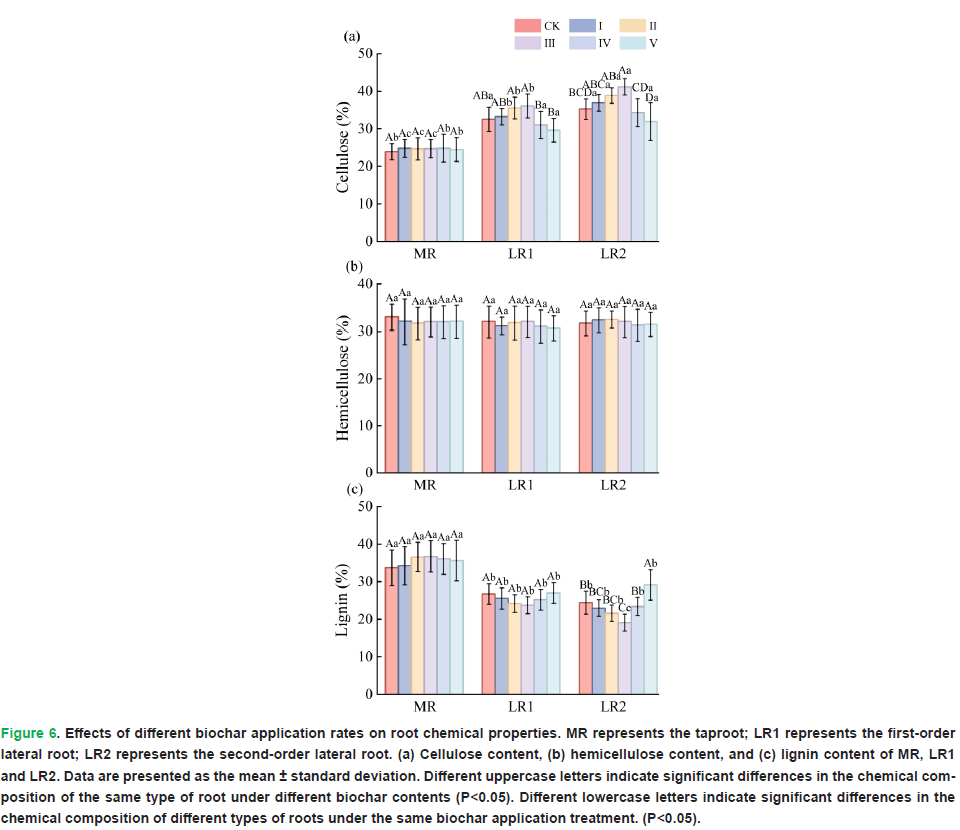
Under different biochar application rates, no significant differences were observed in the cellulose content (P=0.985), hemicellulose content (P=0.979), and lignin content (P=0.650) of the taproots. With respect to first-order lateral roots, the biochar application rate significantly affected the cellulose content (P=0.000), whereas the hemicellulose content (P=0.895) and lignin content (P=0.064) were not significantly influenced. In contrast, for second-order lateral roots, both the cellulose content (P=0.000) and lignin content (P=0.000) were significantly affected by the biochar application rate, whereas the hemicellulose content (P=0.922) was not significantly affected (Fig. 6).
Figure 6: Effects of different biochar application rates on root chemical properties. MR represents the taproot; LR1 represents the first-order lateral root; LR2 represents the second-order lateral root. (a) Cellulose content, (b) hemicellulose content, and (c) lignin content of MR, LR1 and LR2. Data are presented as the mean ± standard deviation. Different uppercase letters indicate significant differences in the chemical composition of the same type of root under different biochar contents (P<0.05). Different lowercase letters indicate significant differences in the chemical composition of different types of roots under the same biochar application treatment. (P<0.05).
There were also differences in the chemical composition among different root types. Under the same treatment, the cellulose content in taproots was lower than that in first-order and second-order lateral roots, whereas the lignin content was higher than that in first-order and second-order lateral roots. No significant differences were observed in hemicellulose content among different root types under the same biochar application rate.
Root tensile strength
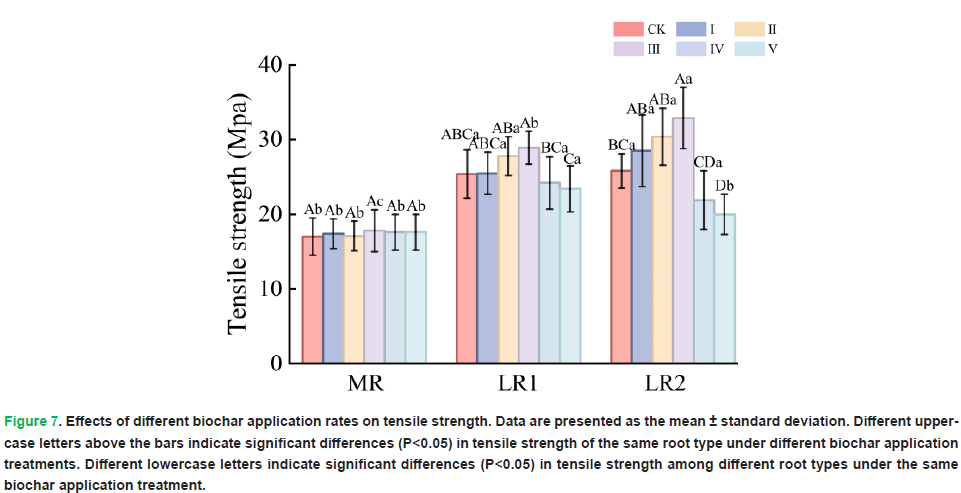
Different biochar application rates did not significantly affect the tensile strength of the taproots (P=0.980). In contrast, significant differences in tensile strength were observed between first-order lateral roots (P=0.001) and second-order lateral roots (P=0.001) across the different treatments. Compared with that in the other treatments, the root tensile strength in Treatment III was significantly greater. Additionally, the tensile strength of the taproots was lower than that of the first-order lateral roots and second-order lateral roots (Fig. 7).
Figure 7: Effects of different biochar application rates on tensile strength. Data are presented as the mean ± standard deviation. Different uppercase letters above the bars indicate significant differences (P<0.05) in tensile strength of the same root type under different biochar application treatments. Different lowercase letters indicate significant differences (P<0.05) in tensile strength among different root types under the same biochar application treatment.
Relationships between root tensile strength and root chemical composition
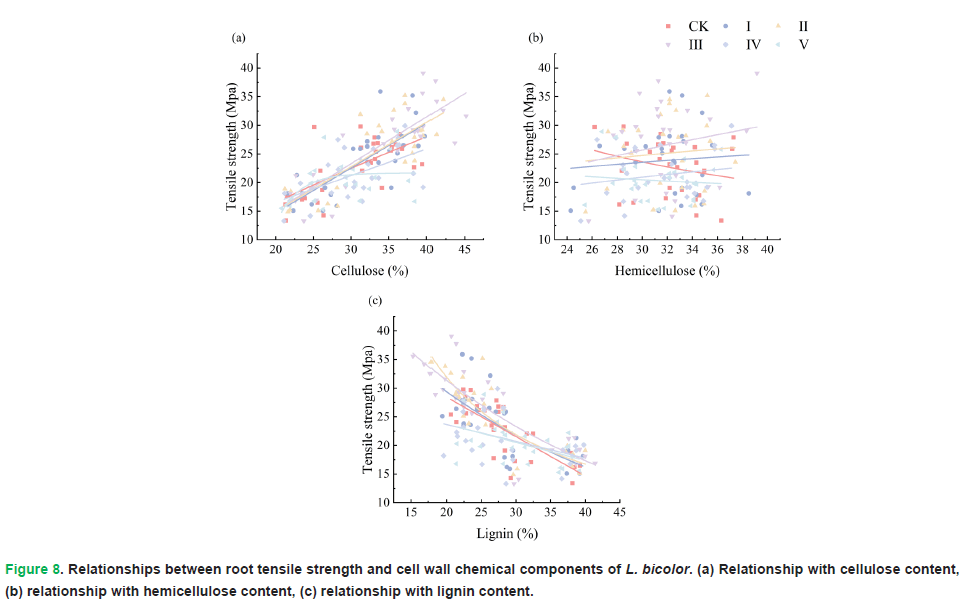
The quantitative relationships between root tensile strength and root chemical composition in each treatment are shown in Fig. 8. Root tensile strength exhibited an increasing trend with increasing cellulose content, and a decreasing trend with increasing lignin content. In other words, root tensile strength was positively correlated with cellulose content and negatively correlated with lignin content. However, no obvious trend was observed between root tensile strength and hemicellulose content (Tab. 2).
Figure 8: Relationships between root tensile strength and cell wall chemical components of L. bicolor. (a) Relationship with cellulose content, (b) relationship with hemicellulose content, (c) relationship with lignin content.
| Root chemical compositions | Treatments | Tensile strength (Mpa) | |
|---|---|---|---|
| Equation | R2 | ||
| Cellulose | Control (CK) | y=1.70x0.76 | 0.45** |
| I | y=0.78x-0.95 | 0.57** | |
| II | y=0.77x-0.45 | 0.63** | |
| III | y=0.82x-1.15 | 0.71** | |
| IV | y=0.46x+7.34 | 0.33** | |
| V | y=21.67-7656.40 × 0.71x | 0.26* | |
| Hemicellulose | Control (CK) | y=175.63x-0.59 | 0.06** |
| I | y=11.38x0.21 | 0.01** | |
| II | y=10.62x0.25 | 0.01** | |
| III | y=3.99x0.55 | 0.04** | |
| IV | y=6.48x0.34 | 0.03** | |
| V | y=37.01x-0.17 | 0.01** | |
| Lignin | Control (CK) | y=-0.69x+42.26 | 0.57** |
| I | y=53.16e-0.03x | 0.46** | |
| II | y=521.20x-0.93 | 0.73** | |
| III | y=56.79e-0.03x | 0.67** | |
| IV | y=-0.29x+29.55 | 0.20* | |
| V | y=-0.32x+30.16 | 0.24** | |
| Note: The equations in the table were fitted using all root types within the same treatment. * denotes a significant correlation at the 0.05 level. ** denotes a highly significant correlation at the 0.01 level. | |||
Table 2. Regression equations between root tensile strength and root chemical compositions of L. bicolor.
Discussion
Effects of biochar application rate on the growth of L. bicolor
Biochar application rate had a significant dose effect on plant growth. Appropriate biochar application could create favorable conditions for plant growth by optimizing the soil microenvironment, whereas excessive addition would disrupt the ecological balance and thus inhibit plant growth. In the present experiment, Treatment III achieved a better balance between soil nutrient supply and plant uptake.
Compared with Control (CK), under the low biochar application rate groups (Treatments I, II, and III), biochar could improve soil health and enhance the plant’s absorption capacity for nitrogen, phosphorus, and potassium, thereby providing favorable conditions for plant growth (Singh, et al. 2015). Conversely, excessive biochar application can temporarily reduce the availability of soil mineral nutrients (Tammeorg, et al. 2014) and disrupt soil microbial biomass and activity (Ameloot, et al. 2014). Such detrimental effects ultimately impede plant growth, leading to significant reductions in both plant height and basal diameter.
Under the influence of the aforementioned regulatory mechanisms, both plant height and basal diameter in the present study exhibited a trend of increasing initially and then decreasing with escalating biochar application rate. Consistent with this observation, Yang, et al. 2024 also confirmed a similar pattern in flue-cured tobacco, where plant height first increased and subsequently decreased as biochar application rate rose, which aligns well with our experimental results. Similarly, Cai, et al. 2021 reported that biochar can significantly promote plant growth and biomass accumulation at an appropriate application rate, whereas an inhibitory effect is induced when the application rate exceeds a critical threshold.
In our experiment, the number of L. bicolor plants per unit area increased continuously with increasing biochar application rate. The main reason was the sustained improvement of soil conditions by biochar. The soil at the experimental site was infertile, and biochar application alleviated adverse environmental conditions such as soil acid-base imbalance and salt stress, thereby creating a more favorable microenvironment for seed germination and seedling survival of L. bicolor (Zulfiqar, et al. 2022, Su, et al. 2024, Antonangelo, et al. 2025). As an inert carbonaceous material, biochar did not promote plant growth through direct nutrient supply at high application rates. Instead, it increased population quantity by reducing the seedling missing rate and seedling mortality. Consequently, the number of L. bicolor plants per unit area continued to rise with increasing biochar application rate.
Notably, the individual growth of L. bicolor showed a threshold effect with increasing biochar application rate, while its population quantity continued to increase. The soil amelioration effect of biochar preferentially supported seed germination and seedling survival, rather than rapid individual growth (Marsh, et al. 2023). When the application rate exceeded the threshold, negative effects such as soil nutrient immobilization only inhibited individual growth, without offsetting the increase in population quantity (Laghari, et al. 2015).
Effects of biochar application rate on root morphology
As a critical organ for water and nutrient uptake, plant roots exhibit strong environmental adaptability (Montagnoli, 2022). Root biomass, as an important indicator of plant growth, is significantly influenced by external environmental conditions during its formation and accumulation (Helmisaari, et al. 2007). Due to its unique microporous structure (Atkinson, et al. 2010, Igalavithana, et al. 2017), biochar exerts multiple beneficial effects in soil amelioration: it can not only effectively increase soil organic matter (Pan, et al. 2009) but also possess excellent water and nutrient retention capacities (Laird, et al. 2010, Ahmad, et al. 2014), thereby significantly improving soil fertility (Mohamed, et al. 2016).
The results of this study indicate that suitable biochar application rates (Treatments I, II, and III) significantly promoted root biomass accumulation (Zou, et al. 2021). On the one hand, biochar addition significantly increases soil organic carbon content (Novak, et al. 2009, Dong, et al. 2018), enhances soil water-holding capacity and water regulation function, and improves soil physical structure by increasing porosity and aeration (Busscher, et al. 2010), thus creating a favorable environment for root growth. On the other hand, biochar can increase soil enzyme activity and the abundance of soil microbial communities, in turn increasing root activity.
In this study, root length, specific root length, and taproot diameter all showed a trend of increasing first and then decreasing, reaching the peak under Treatment III. As a porous material, biochar reduces soil bulk density and increases soil porosity when applied to soil (Jiang, et al. 2025). This modification positively affects root development, resulting in longer root systems. At the optimal application rate, biochar creates a more favorable growth environment for roots by improving soil physical structure, increasing water and nutrient retention capacity, and regulating the pH of acidic soils (Lee, et al. 2025). However, when the application rate exceeds this threshold, the strong adsorption capacity of biochar may cause excessive immobilization of soil nutrients, particularly nitrogen, thereby restricting nutrient acquisition by plants (Zheng, et al. 2013, Prapagdee, et al. 2014). In addition, excessive biochar application may lead to soil loosening, which impairs the contact between roots and soil particles and has complex effects on the structure and function of soil microbial communities, indirectly interfering with the normal development of roots. These adaptive changes in root morphology may help maximize surface area and nutrient absorption, thereby potentially increasing root growth rates.
Biochar application optimizes root morphology and thereby exerts a soil reinforcement effect. Plants under an appropriate biochar application rate present superior root morphological characteristics, such as greater total root length (Chang, et al. 2021), which provides critical support for the improved soil erosion resistance in this study. In terms of soil reinforcement mechanisms, longer total root length can not only expand the root-soil contact area (Blume-Werry, et al. 2019) and strengthen the entanglement and fixation between soil particles (Li, et al. 2020), but also further improve soil structure and enhance soil aggregate stability through root exudates (Lu, et al. 2020, Xiao, et al. 2024). An increase in total root length also indicates a more developed lateral root system. Well-developed lateral roots enhance slope stability and reduce the risk of shallow landslides via reinforcement and anchorage effects (Gray and Barker, 2004, Schwarz, et al. 2010). These results are consistent with previous studies: soil erosion resistance is directly determined by root biomass and morphological characteristics, and is positively correlated with root biomass, taproot length and root diameter. Yang, et al. 2021 further confirmed that root pull-out resistance, as an important indicator of soil erosion resistance, is closely related to key root morphological indices including root diameter, root length and root surface area. For L. bicolor, Zhou, et al. 2023 also clearly demonstrated that root biomass and morphological characteristics play a crucial role in strengthening soil anti-scourability. Accordingly, appropriate biochar amendment optimizes root morphology, and combined with the inherent root advantages of Lespedeza, ultimately leads to a significant improvement in soil erosion resistance.
Effects of biochar application rate on root tensile strength and chemical composition
The ability of plants to resist soil and water loss depends on root tensile properties. In the present study, the tensile strength of taproots showed no significant change, while that of lateral roots increased with root order. The overall tensile strength of the root system initially increased and then decreased with increasing biochar application rate (Fig. 7). Root presence influences the apparent cohesion of soil, and its reinforcement effect plays an important role in the overall shear strength of soil (Ali and Osman 2008, Pallewattha, et al. 2019). Moreover, the improvement in soil cohesion and shear strength is closely related to root tensile strength (Ali and Osman, 2008, Lin, et al. 2024). Therefore, the application of an appropriate amount of biochar can improve the tensile properties of plant roots, leading to a more prominent effect in root-reinforced soil for soil and water conservation.
Root tensile properties serve as a pivotal indicator for assessing soil and water conservation capacity as well as slope and bank stabilization performance. Our findings demonstrate that root tensile strength is intimately associated with root chemical composition. As the primary structural component of plant cell walls (Haigler, et al. 2001), cellulose polymerizes into microfibrils that form the structural framework of root cell walls (Anderson, et al. 2010), thereby governing the mechanical characteristics of plant organs (Bidhendi and Geitmann, 2016). Notably, cellulose microfibrils exhibit remarkable resistance to tensile failure; consequently, elevated cellulose content can substantially enhance root tensile strength. In contrast to cellulose, hemicellulose features a branched structure and functions as a filling and cross-linking component in plant cell walls, improving cell wall flexibility. Lignin represents another critical structural constituent of plant cell walls (Khan, et al. 2024). It interacts with cellulose through hemicellulose-mediated bridges, reinforcing cell wall integrity and augmenting root tensile resistance (Ye, et al. 2017). However, excessive lignin compromises cell wall elasticity while enhancing rigidity, rendering roots more susceptible to brittle fracture under tensile stress (Qi, et al. 2024). This phenomenon was corroborated in Yan, et al. 2022 study on the roots of Dolichos lablab L., a leguminous herbaceous species. Consistent with prior research (Genet, et al. 2005, Hales, et al. 2009), the current study reveals a significant positive correlation between tensile strength and cellulose content, alongside a significant negative correlation between tensile strength and lignin content (Fig. 8). Furthermore, Figs. 6 and 7 provide direct visual evidence that roots with higher tensile strength typically correspond to higher cellulose levels and lower lignin contents.
This study further analyzed the regulatory effects of biochar application rates on root chemical constituents. As illustrated in Fig. 6, for second-order lateral roots, an optimal biochar application rate significantly increased the root cellulose content while reducing the lignin content, which was consistent with the variation trend of the root tensile strength. This suggests that biochar may modulate the synthesis and deposition of root cell wall components (Ali, et al. 2022, Xu, et al. 2022) by improving the rhizosphere microenvironment and regulating carbon metabolic pathways (Kolton, et al. 2017, Jin, et al. 2024), thereby enhancing root mechanical properties from the perspective of chemical composition and providing support for improved root erosion resistance.
Conclusion
These results indicate that biochar plays a crucial role in regulating the soil-reinforcement capacity of the roots of L. bicolor. The addition of an optimal amount of biochar increases root biomass and lateral root cellulose content, decreases lignin content, promotes root development, and thereby significantly increases root tensile strength. In contrast, excessive biochar application has negative effects on root growth. Root tensile strength is positively correlated with the cellulose content and negatively correlated with the lignin content. Based on the findings of this study, a biochar application rate of 7.5 kg·m-2 is deemed suitable for improving the soil-reinforcement capacity of L. bicolor roots in practical applications. Future research should further investigate the long-term impacts of biochar across a broader range of application rates and plant species to provide a theoretical basis for ecological restoration projects.
Acknowledgments
We thank the National Natural Science Foundation of China (42277162 and U22A20601) and Sichuan Transportation Science and Technology Program (2018-ZL-15 and 2022-A-5) for their support. We also thank the anonymous reviewers for their valuable comments and suggestions, which helped improve the quality of this paper.Competing Interests
The authors declare that they have no conflict of interest.
Data Availability
The data are available from the authors upon reasonable request.
References
- Ahmad M, Rajapaksha AU, Lim JE, Zhang M, Bolan N, Mohan D, Vithanage M, Lee SS, Ok YS. (2014). Biochar as a sorbent for contaminant management in soil and water: A review. Chemosphere. 99:19-33.
[Crossref] [Google Scholar] [PubMed]
- Ali FH, Osman N. (2008). Shear strength of a soil containing vegetation roots. Soils and Foundations. 48:587-596.
- Ali I, Adnan M, Iqbal A, Ullah S, Khan MR, Yuan P, Zhang H, Nasar J, Gu M, et al. (2022). Effects of biochar and nitrogen application on rice biomass saccharification, bioethanol yield and cell wall polymers features. Int J Mol Sci. 23:13635.
- Ameloot N, Sleutel S, Case SDC, Alberti G, McNamara NP, Zavalloni C, Vervisch B, Vedove Gd, De Neve S. (2014). C mineralization and microbial activity in four biochar field experiments several years after incorporation. Soil Biol Biochem. 78:195-203.
- Antonangelo JA, Sun X, Eufrade-Junior HD. (2025). Biochar impact on soil health and tree-based crops: A review. Biochar. 7:51.
- Anderson CT, Carroll A, Akhmetova L, Somerville C. (2010). Real-time imaging of cellulose reorientation during cell wall expansion in Arabidopsis roots. Plant Physiol. 152:787-796.
[Crossref] [Google Scholar] [PubMed]
- Atkinson CJ, Fitzgerald JD, Hipps NA. (2010). Potential mechanisms for achieving agricultural benefits from biochar application to temperate soils: A review. Plant and Soil. 337:1-18.
- Barberis D, Lombardi G, Ravetto Enri S, Pittarello M, Viglietti D, Freppaz M, Lonati M. (2023). Nitrogen fertilizer enhances vegetation establishment of a high-altitude machine-graded ski slope. Restor Ecol. 31:e13777.
- Bezzalla A, Bennadji MEA, Zidane L, Neffar S, Chenchouni H. (2026). A review on the impact of biochar applications on soil health and fertility, plant growth, and food security: Advancing agricultural sustainability. Biomass Bioenerg. 209:108960.
- Bidhendi AJ, Geitmann A. (2016). Relating the mechanics of the primary plant cell wall to morphogenesis. J Exp Bot. 67:449-461.
[Crossref] [Google Scholar] [PubMed]
- Blume-Werry G, Milbau A, Teuber LM, Johansson M, Dorrepaal E. (2019). Dwelling in the deep-strongly increased root growth and rooting depth enhance plant interactions with thawing permafrost soil. New Phytol. 223:1328-1339.
[Crossref] [Google Scholar] [PubMed]
- Busscher WJ, Novak JM, Evans DE, Watts DW, Niandou MAS, Ahmedna M. (2010). Influence of pecan biochar on physical properties of a Norfolk loamy sand. Soil Sci. 175:10-14.
- Cai JF, Fan J, Liu XS, Sun K, Wang W, Zhang MX, Li HL, Xu HF, Kong WJ, et al. 2021. Biochar-amended coastal wetland soil enhances growth of Suaeda salsa and alters rhizosphere soil nutrients and microbial communities. Sci Total Environ. 788:147707.
[Crossref] [Google Scholar] [PubMed]
- Chang YR, Rossi L, Zotarelli L, Gao B, Shahid MA, Sarkhosh A. 2021. Biochar improves soil physical characteristics and strengthens root architecture in Muscadine grape (Vitis rotundifolia L.). Chem Biol Technol Agric. 8:7.
- Chen QY, Xiao SL, Shi SQ, Cai LP. (2018). Isolation of cellulose from poplar wood by nitric acid-ethanol treatment and its effect on the quality of films cast from ionic liquid. Bioresour. 13:8943-8955.
- Coomes OT, Miltner BC. (2017). Indigenous charcoal and biochar production: Potential for soil improvement under shifting cultivation systems. Land Degrad Dev. 28:811-821.
- De Baets S, Poesen J. (2010). Empirical models for predicting the erosion-reducing effects of plant roots during concentrated flow erosion. Geomorphology. 118:425-432.
- Dong XL, Singh BP, Li GT, Lin QM, Zhao XR. (2018). Biochar application constrained native soil organic carbon accumulation from wheat residue inputs in a long-term wheat-maize cropping system. Agric Ecosyst Environ. 252:200-207.
- Dupuy L, Fourcaud T, Stokes A. (2005). A numerical investigation into the influence of soil type and root architecture on tree anchorage. Plant and Soil. 278:119-134.
- Eab KH, Likitlersuang S, Takahashi A. (2015). Laboratory and modelling investigation of root-reinforced system for slope stabilisation. Soil Found. 55:1270-1281.
- El-Naggar A, Lee SS, Awad YM, Yang X, Ryu C, Rizwan M, Rinklebe J, Tsang DCW, Ok YS. (2018). Influence of soil properties and feedstocks on biochar potential for carbon mineralization and improvement of infertile soils. Geoderma. 332:100-108.
- Genet M, Stokes A, Salin F, Mickovski SB, Fourcaud T, Dumail J-F, van Beek R. (2005). The influence of cellulose content on tensile strength in tree roots. Plant and Soil. 278:1-9.
- Gray DH, Barker D. (2004). Root-soil mechanics and interactions. Riparian Vegetation and Fluvial Geomorphology. 8:113-123.
- Haigler CH, Ivanova-Datcheva M, Hogan PS, Salnikov VV, Hwang S, Martin K, Delmer DP. (2001). Carbon partitioning to cellulose synthesis. Plant Mol Biol. 47:29-51.
[Crossref] [Google Scholar] [PubMed]
- Hales TC, Ford CR, Hwang T, Vose JM, Band LE. (2009). Topographic and ecologic controls on root reinforcement. J Geophys Res Earth Surf. 114:F03013.
- Helmisaari H-S, Derome J, Nöjd P, Kukkola M. (2007). Fine root biomass in relation to site and stand characteristics in Norway spruce and Scots pine stands. Tree Physiol. 27:1493-1504.
[Crossref] [Google Scholar] [PubMed]
- Igalavithana AD, Ok YS, Niazi NK, Rizwan M, Al-Wabel MI, Usman ARA, Moon DH, Lee SS. (2017). Effect of corn residue biochar on the hydraulic properties of sandy loam soil. Sustainability. 9:266.
- Isimikalu TO, Olaniyan JO, Affinnih KO, Muhammed OA, Adede AC, Jibril AH, Atteh E, Yusuf S, Juliana Ezekiel T. (2023). Rice husk biochar and inorganic fertilizer amendment combination improved the yield of upland rice in typical soils of Southern Guinea Savannah of Nigeria. Int J Recycl Org Waste Agric. 12:1-16.
- Jiang Z, Huang S, Meng ZW. (2025). Long-term effects of biochar on the hydraulic properties of soil: A meta-analysis based on 1-10 years field experiments. Geoderma. 458:117318.
- Jin W, Liu ZT, Cheng ZR, Wang Q, Zhang Y, Hu W, Chen BL, Zhou ZG, Meng YL. (2024). External carbon addition alters soil photosynthetic carbon accumulation and rhizosphere processes of carbon. Appl Soil Ecol. 201:105478.
- Khan GA, Dutta A, van de Meene A, Frandsen KEH, Ogden M, Whelan J, Persson S. (2024). Phosphate starvation regulates cellulose synthesis to modify root growth. Plant Physiol. 194:1204-1217.
[Crossref] [Google Scholar] [PubMed]
- Kolton M, Graber ER, Tsehansky L, Elad Y, Cytryn E. (2017). Biochar-stimulated plant performance is strongly linked to microbial diversity and metabolic potential in the rhizosphere. New Phytol. 213:1393-1404.
[Crossref] [Google Scholar] [PubMed]
- Laghari M, Mirjat MS, Hu Z, Fazal S, Xiao B, Hu M, Chen Z, Guo D. (2015). Effects of biochar application rate on sandy desert soil properties and sorghum growth. Catena. 135:313-320.
- Laird D, Fleming P, Wang BQ, Horton R, Karlen D. (2010). Biochar impact on nutrient leaching from a Midwestern agricultural soil. Geoderma. 158:436-442.
- Lee SH, Park MW, Chang H, Je SM, Kim GJ, Noh NJ. (2025). Effects of soil physical ameliorants on the growth and root morphology of Prunus yedoensis and Ginkgo biloba seedlings in compacted soils. J For Res. 30:262-271.
- Li JY, Chen QF, Li Q, Zhao CS, Feng Y. (2021). Influence of plants and environmental variables on the diversity of soil microbial communities in the Yellow River Delta Wetland, China. Chemosphere. 274:129967.
[Crossref] [Google Scholar] [PubMed]
- Li JY, Yuan XL, Ge L, Li Q, Li ZG, Wang L, Liu Y. (2020). Rhizosphere effects promote soil aggregate stability and associated organic carbon sequestration in rocky areas of desertification. Agric Ecosyst Environ. 304:107126.
- Li ZJ, Xiang JM, Zhuo GC, Zhang HY, Dai KR, Shi XL. (2025). Dynamic landslide susceptibility assessment in the Yalong river alpine gorge region integrating InSAR-derived deformation velocity. Remote Sensing. 17:3210.
- Lin YZ, Jian WB, Lai ZR, Zhong X, Zhang JY, Xia C. (2024). Mechanical properties of root-soil composite in tree-covered landslide area based on field prototype test. Rock Soil Mech. 45:3423-3434.
- Lu JR, Zhang Q, Werner AD, Li YL, Jiang SY, Tan ZQ. (2020). Root-induced changes of soil hydraulic properties-A review. J Hydrol. 589: 125203.
- Ma JY, Li ZB, Li P, Ma B, Xiao L, Cui ZW, Wang Z, Min ZQ. (2024). Effect of mixed plant roots on saturated hydraulic conductivity and saturated water content of soil in the loess region. Agric Water Manag. 295:108784.
- Major J, Rondon M, Molina D, Riha SJ, Lehmann J. (2010). Maize yield and nutrition during 4 years after biochar application to a Colombian savanna oxisol. Plant and Soil. 333:117-128.
- Marsh C, Blankinship JC, Hurteau MD. (2023). Effects of nurse shrubs and biochar on planted conifer seedling survival and growth in a high-severity burn patch in New Mexico, USA. For Ecol Manage. 537:120971.
- Mohamed BA, Ellis N, Kim CS, Bi X, Emam AE. (2016). Engineered biochar from microwave-assisted catalytic pyrolysis of switchgrass for increasing water-holding capacity and fertility of sandy soil. Sci Total Environ. 566:387-397.
- Montagnoli A. (2022). Adaptation of the root system to the environment. Forests. 13:595.
- Murtaza G, Ahmed Z, Eldin SM, Ali B, Bawazeer S, Usman M, Iqbal R, Neupane D, Ullah A, Khan A, Hassan MU. (2023). Biochar-soil-plant interactions: A cross talk for sustainable agriculture under changing climate. Front Mar Sci. 11:1059449.
- Murtaza G, Ahmed Z, Usman M, Tariq W, Ullah Z, Shareef M, Iqbal H, Waqas M, Tariq A, Wu Y, Zhang Z. (2021). Biochar induced modifications in soil properties and its impacts on crop growth and production. J Plant Nutr. 44:1677-1691.
- Novak JM, Busscher WJ, Laird DL, Ahmedna M, Watts DW, Niandou MAS. (2009). Impact of biochar amendment on fertility of a southeastern coastal plain soil. Soil Sci. 174:105-112.
- Oram NJ, van de Voorde TFJ, Ouwehand G-J, Bezemer TM, Mommer L, Jeffery S, Groenigen JWV. (2014). Soil amendment with biochar increases the competitive ability of legumes via increased potassium availability. Agric Ecosyst Environ. 191:92-98.
- Pan GX, Smith P, Pan WN. (2009). The role of soil organic matter in maintaining the productivity and yield stability of cereals in China. Agric Ecosyst Environ. 129:344-348.
- Pallewattha M, Indraratna B, Heitor A, Rujikiatkamjorn C. (2019). Shear strength of a vegetated soil incorporating both root reinforcement and suction. Transp Geotech. 18:72-82.
- Prapagdee S, Piyatiratitivorakul S, Petsom A, Tawinteung N. (2014). Application of biochar for enhancing cadmium and zinc phytostabilization in Vigna radiata L. cultivation. Water Air Soil Poll. 225:2233.
- Qi M, Li MH, Wang XW, Song YY, Sun HL. (2024). Root morphological and mechanical properties of Lespedeza bicolor Turcz. on slopes at different altitudes. Trees. 39:6.
- Sadaf J, Shah GA, Shahzad K, Ali N, Shahid M, Ali S, Hussain RA, Ahmed ZI, Traore B, Ismail IM, Rashid MI. (2017). Improvements in wheat productivity and soil quality can accomplish by co-application of biochars and chemical fertilizers. Sci Total Environ. 607:715-724.
[Crossref] [Google Scholar] [PubMed]
- Schwarz M, Lehmann P, Or D. (2010). Quantifying lateral root reinforcement in steep slopes-from a bundle of roots to tree stands. Earth Surf Process Landforms. 35:354-367.
- Scrimgeour C. (2008). Soil sampling and methods of analysis (second edition). Exp Agric. 44:437-437.
- Sharma P. (2024). Biochar application for sustainable soil erosion control: A review of current research and future perspectives. Front Env Sci. 12:1-12.
- Singh R, Babu JN, Kumar R, Srivastava P, Singh P, Raghubanshi AS. (2015). Multifaceted application of crop residue biochar as a tool for sustainable agriculture: An ecological perspective. Ecol Eng. 77:324-347.
- Su Z, Liu X, Wang Z, Wang J. (2024). Biochar effects on salt-affected soil properties and plant productivity: A global meta-analysis. J Environ Manage. 366:121653.
[Crossref] [Google Scholar] [PubMed]
- Sun DY, Li JZ, Gong YB. (2023). Effects of planting density of Poaceae species on slope community characteristics and artificial soil nutrients in high-altitude areas. Sustainability. 15:8321.
- Tammeorg P, Simojoki A, Mäkelä P, Stoddard FL, Alakukku L, Helenius J. (2014). Short-term effects of biochar on soil properties and wheat yield formation with meat bone meal and inorganic fertiliser on a boreal loamy sand. Agric Ecosyst Environ. 191:108-116.
- Wang M, Lin M, Liu QH, Zhang Y, Luo R, Pang XY. (2024). Altitudinal decline of vegetation restoration effects on soil microbial communities on high-altitude roadside slopes: environmental drivers and management implications. Sci Total Environ. 957:177424.
[Crossref] [Google Scholar] [PubMed]
- Xiang YZ, Deng Q, Duan HL, Guo Y. (2017). Effects of biochar application on root traits: a meta-analysis. Global Change Biology Bioenergy. 9:1563-1572.
- Xiao T, Li P, Fei WB, Wang JD. (2024). Effects of vegetation roots on the structure and hydraulic properties of soils: A perspective review. Sci Total Environ. 906:167524.
[Crossref] [Google Scholar] [PubMed]
- Xiong SM, Zuo XF, Zhu YY. (2005). Determination of Cellulose, Hemi-Cellulose and Lignin in Rice Hull. Cereal & Feed Industry. 40-41.
- Xu M, Gao P, Wu J, Ma J, Zhang XH, Yang G, Long LL, Chen C, Song C, Xiao Y. (2022). Biochar promotes arsenic sequestration on iron plaques and cell walls in rice roots. Chemosphere. 288:132422.
[Crossref] [Google Scholar] [PubMed]
- Yan ZH, Chen Y, Liu XH, He BH, Tang H, Qiang JJ. (2022). Comprehensive evaluation of shear strength and anti-scourability of root-soil complex of two grass hedgerows in karst slope land. Acta Ecologica Sinica. 42:1811-1820.
- Yang QH, Zhang CB, Liu PC, Jiang J. (2021). The role of root morphology and pulling direction in pullout resistance of alfalfa roots. Front Plant Sci. 12:580825.
[Crossref] [Google Scholar] [PubMed]
- Yang YF, Ahmed W, Ye CH, Yang LY, Wu LZ, Dai ZL, Khan KA, Hu XD, Zhu XH, Zhao Z. (2024). Exploring the effect of different application rates of biochar on the accumulation of nutrients and growth of flue-cured tobacco (Nicotiana tabacum). Front Plant Sci. 15:1225031.
[Crossref] [Google Scholar] [PubMed]
- Ye C, Guo ZL, Li ZX, Cai CF. (2017). The effect of Bahiagrass roots on soil erosion resistance of Aquults in subtropical China. Geomorphology. 285:82-93.
- Zanati MR, Guirguis MA, Saber MSM. (1973). Biological and chemical determination of available potassium in soil. Zentralbl Bakteriol Parasitenkd Infektionskr Hyg. 128:572-577.
[Crossref] [Google Scholar] [PubMed]
- Zhang CB, Chen LH, Jiang J. (2014). Why fine tree roots are stronger than thicker roots: The role of cellulose and lignin in relation to slope stability. Geomorphology. 206:196-202.
- Zheng H, Wang ZY, Deng X, Herbert S, Xing BS. (2013). Impacts of adding biochar on nitrogen retention and bioavailability in agricultural soil. Geoderma. 206:32-39.
- Zhou MX, Yan GY, Li YB, Chen D, Yan C, Wang N, Jia C, Wang QG, Xing YJ. (2023). Root biomass and root morphological traits of three shrub species: Implications for soil anti-scouring resistance of ecological slopes. PLoS One. 18:e0288848.
[Crossref] [Google Scholar] [PubMed]
- Zou ZH, Fan LC, Li X, Dong CW, Zhang LP, Zhang L, Fu JY, Han WY, Yan P. (2021). Response of plant root growth to biochar amendment: a meta-analysis. Agronomy. 11:2442.