Research Article - Modern Phytomorphology ( 2026) Volume 20, Issue 1
Development and characterization of a functional yogurt fortified with Moringa oleifera freeze dried extract powder
Maryam Saeed1, Shinawar Waseem Ali1*, Muhammad Rizwan Tariq1, Mateen Ahmad2, Muzzamal Ahmed Muzzafar1, Kainat Ashfaq1, Muhammad Riaz3, Aftab Ahmad4, Hafiz Roy Umair5 and Qurban Ali62Department of Human Nutrition and Dietetics, Gulab Devi Educational Complex, Lahore, Pakistan
3Department of Food Safety and Quality Management, Bahauddin Zakariya University, Multan, Pakistan
4Department of Nutritional Sciences, Government College University, Faisalabad, Pakistan
5Fahmir Pharma Private Limited Industry, Shehikupura, Pakistan
6Department of Plant Breeding & Genetics, Faculty of Agricultural Sciences, University of the Punjab, Lahore, Pakistan
Shinawar Waseem Ali, Department of Food Sciences, Faculty of Agricultural Sciences, University of the Punjab, Lahore, Pakistan, Email: shinawar.foodsciences@pu.edu.pk
Received: 06-Feb-2026, Manuscript No. mp-26-184002; Accepted: 01-Apr-2026, Pre QC No. mp-26-184002 (PQ); Editor assigned: 09-Feb-2026, Pre QC No. mp-26-184002 (PQ); Reviewed: 23-Feb-2026, QC No. mp-26-184002; Revised: 31-Mar-2026, Manuscript No. mp-26-184002 (R); Published: 08-Apr-2026, DOI: 10.5281/zenodo.19624762
Abstract
Background: Moringa oleifera demonstrates substantial potential as a valuable ingredient in the food industry due to its nutrient- dense composition, including high protein levels, vitamins, and minerals. This study analyzed Moringa Freeze Dried Extract Powder (MFDP) fortified yogurt for its composition, benefits, quality, and sensory characteristics. Notably, incorporating MFDP into yogurt offers a promising functional food approach to support glycemic control and overall metabolic health.
Materials and methods: The MFDP was incorporated into yogurt in four different concentration 0.5% (T1), 1% (T2), 1.5% (T3), and 2% (T4) and observed for its quality and functional properties over a 15-day storage period at 4 ± 1ºC. The study assessed nutritional, physicochemical properties, antioxidant potential, alpha amylase inhibition activity, and microstructural characteristics of yogurt. Additionally, the study assessed the viability of yogurt starter culture Streptococcus thermophilus and Lactobacillus bulgaricus across all treatment groups to determine the impact of MFDP on microbial growth survival.
Results and discussion: Results demonstrated that MFDP yogurt maintained the stability of bioactive compounds, with notable levels of vitamin C (8.54-8.60 mg/g), total phenols (18.13-28.13 mg GAE/g), and total flavonoids (17.52-28.52 mg RE/100 g). The MFDP fortified yogurt showed significantly (p<0.05) decreased pH (4.26-4.17) and moisture (82.16-77.3%), while increasing acidity (1.08-1.13 %) and decreasing syneresis (1.45-1.30 ml) in fortified samples. Moreover, MFDP improved the color scores of the yogurt sample, i.e., a* (-7.99 to -12.48), b* (9.4-11.20), and L (85.19-80.69). The anti-diabetic, anti-oxidative potential was evidenced by increased α-amylase activity (44.80-83.35%). Sensory evaluation indicated enhanced aroma and smoother texture in yogurt with 1.5% (T3) MFDP concentration. Importantly it also contributed additional essential minerals-such as calcium, iron and potassium, not inherently abundant in conventional yogurt, and their concentrations remained significant after fortification. Furthermore, starter culture (Lactobacillus bulgaricus and Streptococcus thermophilus), viability remained stable across treatments, with no adverse effect on fermentation. Microstructural analysis revealed slightly larger particles and smaller pores in fortified yogurt compared to the control. This study underscores Moringa’s promise as a functional ingredient in yogurt, with potential health benefits, particularly for diabetes management and overall well-being.
Conclusion: MFDP-fortified yogurt, particularly at 1.5% concentration, enhances nutritional value, antioxidant activity, and sensory quality while maintaining microbial stability, making it a promising functional food for supporting metabolic health and glycemic control.
Keywords
Moringa oleifera, Antioxidant, Anti-diabetic, Phytonutrients, Aqueous extraction
Abbrevations
MFDP: Moringa Freeze Dried Extract Powder; FRAP: Ferric reducing antioxidant power; SEM: Scanning Electron Microscopy; GAE: Gallic Acid Equivalents; DPPH: 2,2-diphenyl-1-picrylhydrazyl assay; FRAP: Ferric Reducing Antioxidant Power; TPC: Total Phenolic Content; TFC: Total Flavonoid Content; TAC: Total Antioxidant Capacity
Introduction
The global prevalence of Non-Communicable Disease (NCDs), such as diabetes mellitus, is escalating at an alarming rate. According to the international diabetes federation, 2021, more than 530 million adults worldwide currently live with diabetes, and this figure is projected to increase by nearly 46% by 2045. Beyond its direct health impacts, diabetes places a substantial burden on healthcare systems and the economy. Parallel to this, malnutrition and micronutrient deficiencies continue to affect millions, particularly in low and middle-income countries, creating a dual challenge of undernutrition and overnutrition. Addressing such complex health issues requires innovative and affordable dietary strategies that not only meet nutritional requirements but also deliver bioactive compounds with therapeutic potential (Fatima, et al. 2026, Li, et al. 2020). Functional foods have emerged as a promising approach to bridge this gap, offering health benefits beyond basic nutrition. Among various matrices, yogurt is widely recognized as an excellent product for functional food development due to its nutrient density, probiotic content, consumer acceptability and versatility (Granato, et al. 2020, Safdar, et al. 2026). The fortification of yogurt with plant-based bioactive ingredients has been shown to enhance its health-promoting potential, including antioxidant, antimicrobial, and enzyme-inhibitory properties (Bankole, et al. 2023). Such functional enhancement is particularly relevant for combating oxidative stress and hyperglycemia, two key factors implicated in diabetes progression. Moringa oleifera, often referred to as the “miracle tree”, has attracted considerable attention as a nutrient-rich plant source with substantial therapeutic potential. Its leaves are particularly abundant in proteins, essential amino acids, vitamins, minerals, flavonoids and phenolic activities (Lu, et al. 2023, Mehwish, et al. 2022). Evidence suggests that Moringa exerts a-amylase and a-glucosidase inhibitory effects, mimicking mechanisms of conventional antidiabetic drugs, and thus may help attenuate postprandial adaptability and make it highly suitable for incorporation into affordable functional foods, particularly in resource-limited regions.
Despite these promising attributes, the application of Moringa oleifera in dairy-based functional foods remains limited. Previous studies have primarily focused on the use of fresh leaf powder or alcoholic extracts, examining mainly compositional or sensory attributes of fortified yogurts (Vijay, et al. 2022, Sonu, 2023). Although these works demonstrate feasibility, they provide limited insights into the broader functional properties such as bioactive stability during storage, antioxidant capacity, enzyme inhibition, and microbial viability. Importantly, the freeze-drying process, which preserves heat-sensitive vitamins and polyphenols more effectively than traditional drying methods, has rarely been explored in the context of yogurt fortification (Adepoju and Selezneva, 2024). The present study addresses this gap by fortifying yogurt with MFDP and systematically evaluating its impact on nutritional composition, physicochemical parameters, antioxidant activity, a-amylase inhibitory effects, mineral enrichment, sensory acceptability, microbial viability, and microstructural characteristics during refrigerated storage. By integrating these multidimensional assessments, the study not only evaluates the feasibility of MFDP fortified yogurt but also highlights its potential as a timely and globally relevant functional food innovation. Such an approach aligns with the urgent need to develop affordable, sustainable, and evidence-based interventions that can support glycemic control, reduce oxidative stress, and contribute to the prevention of diet-related chronic disease.
Materials and Methods
Raw material procurement
The Moringa oleifera leaf powder was obtained from the private firm Virsa Agri Farms and Services, Faisalabad. The bacterial strains (YOFLEX-Thermophilic Yogurt Culture YC-X11) were procured from CHR Hansen Denmark.
Preparation of Moringa freeze dried extract powder
A fixed ratio of 1:10 (w/v) was employed, wherein 100 g of dried Moringa leaf powder was placed into a borosilicate glass flask, and 1000 mL of distilled water was added. The flask was covered with aluminum foil to minimize evaporation and placed in a temperaturecontrolled water bath maintained at 45-50°C for 75 hours. At the end of the maceration period, the mixture was filtered through Whatman No.1 filter paper (GE Healthcare Life Sciences, Buckinghamshire, UK) to remove plant residues. A rotary evaporator (Tokyo Rikakikai Co., Ltd., Tokyo, Japan) was used to concentrate the filtered solvent, and the residue was freeze-dried by Ilshin Lab Co., Ltd. and kept at -80°C until use. The yield of the MFDP was calculated using the following formula (Bhatta, et al. 2025) (Formula 1).
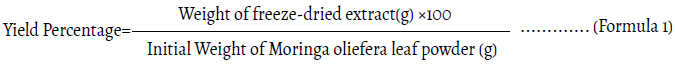
Mother culture preparation and yogurt production
Mother culture was prepared by using 0.2 g/l freeze dried strains of Yoflex (Culture YC-X11), added to pasteurized liquid whole milk, as per the manufacturer’s specifications. The milk was pasteurized at 90° C for 5 minutes, then cooled to 43° C, before inoculation. After the addition of the culture, the milk was incubated at 43°C for 4 hours, and subsequently cooled to 4°C (Dinic, et al. 2023). For yogurt production, the milk was heated at 85°C for 15-20 min, followed by cooling to 40-45°C, then 2.5% (v/v) inoculum was added. Then in four different concentration 0.5% (T1), 1% (T2), 1.5% (T3) and 2% (T4) MFDP were used in making fortified yogurt, while a control sample was prepared without any addition of MFDP. Afterwards it was allowed for incubation at 43 °C for 4 h. These trial yogurt samples were stored at 4°C until further analysis.
Physicochemical characterization of MFDP fortified yogurt
Moisture content was measured by oven-drying samples at 100°C to a constant weight using a hot air oven (Model: DHG-9070A, Yiheng Scientific, China), Crude fat was extracted using a soxhlet appratus (Model: SOX406, Gerhardt, Germany), while crude fiber was analyzed by sequential acid and alkali digestion using a fiber analyzer (Model: F2000, ANOM, USA). Titratable acidity was determined by titrating with 0.1N NaOH using phenolphthalein as an indicator. pH levels were measured using a digital pH meter (Inolab pH 720, WTW 82362), All analyses were conducted in triplicate and recorded in specified units for each parameter. The amount of vitamin C in the MFDP-based yogurt was assessed following the procedure described by Sankhyan, et al. 2013: 300 µL of yogurt was mixed with 100 µl of 13.3% trichloroacetic acid and 100 µl distilled water, followed by 75 µl DNPH reagent (2g DNPH, 230 mg thioureas, 270 mg CuSO4.5H2O in 100 ml 5M H2SO4). After incubation at 37°C for 3 hours, 0.5 ml of 65% H2SO4 was added to terminate the reaction, and absorbance was measured at 520 nm (PerkinElmer Lambda, USA). The results were expressed as Ascorbic Acid Equivalents (AAE) for ascorbic acid (Formula 2).

Mother culture preparation and yogurt production
Mother culture was prepared by using 0.2 g/l freeze dried strains of Yoflex (Culture YC-X11), added to pasteurized liquid whole milk, as per the manufacturer’s specifications. The milk was pasteurized at 90° C for 5 minutes, then cooled to 43° C, before inoculation. After the addition of the culture, the milk was incubated at 43°C for 4 hours, and subsequently cooled to 4°C (Dinic, et al. 2023). For yogurt production, the milk was heated at 85°C for 15-20 min, followed by cooling to 40-45°C, then 2.5% (v/v) inoculum was added. Then in four different concentration 0.5% (T1), 1% (T2), 1.5% (T3) and 2% (T4) MFDP were used in making fortified yogurt, while a control sample was prepared without any addition of MFDP. Afterwards it was allowed for incubation at 43 °C for 4 h. These trial yogurt samples were stored at 4°C until further analysis.
Physicochemical characterization of MFDP fortified yogurt
Moisture content was measured by oven-drying samples at 100°C to a constant weight using a hot air oven (Model: DHG-9070A, Yiheng Scientific, China), Crude fat was extracted using a soxhlet appratus (Model: SOX406, Gerhardt, Germany), while crude fiber was analyzed by sequential acid and alkali digestion using a fiber analyzer (Model: F2000, ANOM, USA). Titratable acidity was determined by titrating with 0.1N NaOH using phenolphthalein as an indicator. pH levels were measured using a digital pH meter (Inolab pH 720, WTW 82362), All analyses were conducted in triplicate and recorded in specified units for each parameter. The amount of vitamin C in the MFDP-based yogurt was assessed following the procedure described by Sankhyan, et al. 2013: 300 µL of yogurt was mixed with 100 µl of 13.3% trichloroacetic acid and 100 µl distilled water, followed by 75 µl DNPH reagent (2g DNPH, 230 mg thioureas, 270 mg CuSO4.5H2O in 100 ml 5M H2SO4). After incubation at 37°C for 3 hours, 0.5 ml of 65% H2SO4 was added to terminate the reaction, and absorbance was measured at 520 nm (PerkinElmer Lambda, USA). The results were expressed as Ascorbic Acid Equivalents (AAE) for ascorbic acid (Formula 2).
Color analysis
A Colorimeter was employed to assess the L*, a*, and b* values of color. The fortified yogurt samples were evaluated for color using a Minolta colorimeter CR-300 calibrated with a white standard porcelain plate. The assessment involved scoring the color based on L* (lightness), a* (redness/greenness), and b* (yellowness/blueness) according to the CIELAB scale (Mousavi, et al. 2019).
Mineral analysis
Due to the high moisture content of yogurt, pre-drying of samples at 105°C was carried out to avoid excessive foaming or spattering during ashing and to facilitate complete removal of water before incineration in the muffle furnace. About 2 g of the dried sample was ashed in a muffle furnace at 550°C for 4 hours. The resulting ash was dissolved in 10 ml of aqua regia (HNO3: HCL=3:1) and diluted to 50 ml with deionized water. The solution was filtered (Whatman No. 42) and analyzed for minerals using an atomic absorption spectrophotometer (PerkinElmer Analyst 400, USA) at specific wavelengths: Ca 422.7nm, Mg 285.2 nm, Zn 213.9 nm, Fe 248.3 nm, and Mn 279.5 nm. Sodium and potassium were determined using a flame photometer (Jenway PFP7) at 598 nm and 766.5 nm, respectively. Phosphorous was measured by the molybdenum blue method at 880 nm, using colorimeter. Results were expressed as mg/100 g of sample (AOAC, 2019).
Determination of antioxidant profile
Two grams of the yogurt sample was homogenized and centrifuged at 4000 rpm for 10 minutes at 4°C, and the obtained supernatant was analyzed to determine the antioxidant profile by using DPPH, FRAP, TPC, TFC and TAC assays (Kim, 2017, Hashemi, et al. 2019). The DPPH radical scavenging activity was determined following Fitriana, et al. 2020, where a 1 mg/ml stock solution of the sample was serially diluted, mixed with 1 ml of freshly prepared 0.1 mM DPPH, incubated for 30 min in the dark and measured at 517 nm; results were expressed as percent inhibition or ascorbic acid equivalents (Equation 3).

Where:
Ac=Absorbance of control
As=Absorbance of sample
FRAP was assessed according to the procedure detailed by Kim, 2020, 150 µl of sample with FRAP reagent (acetate buffer, 10 mM TPTZ in 40 mM HCL, and 20 mM FeCl3.6H2O, 10:1:1), incubating at 37°C for 30 min, and recorded absorbance at 593 nm, with FeSO4.7H2O as the standard. Total phenolic content (TPC) was quantified using the Folic-Ciocalteu method (Stohs and Hartman, 2015), where 0.5 ml of the sample was reacted with Folin-Ciocalteu reagent, and incubated for 30 min before measurement at 755 nm, results expressed as mg Gallic Acid Equivalents (GAE)/100 g. TFC was measured by the NaNO2-AlCl3 colorimetric method (Ghafar, et al. 2017), in which the extract was sequentially treated with NaNO2, AlCl3.6H2O, and adjusted to 3 ml with deionized water, absorbance taken at 510 nm, and results expressed as mg Rutin Equivalents (RE)/g dry weight. Total Antioxidant Capacity (TAC) was estimated using the phosphomolybdenum method (Luqman, et al. 2012), where the sample was incubated with reagent (H2SO4, Na3PO4, (NH4)6Mo.7O24) at 95°C for 90 min, cooled, and measured at 695 nm, expressed as mg Ascorbic Acid Equivalents (AAE)/g.
Alpha amylase inhibition assay
Following Freitas, et al. 2023, 1-2 g of yogurt were extracted with 10 ml methanol, vortexed for 10-15 minutes, centrifuged (5000 rpm, 10 min), and the supernatant filtered (0.45 µm) with a membrane. For the assay, 200 µL alpha-amylase (1U/mL in 20 mM phosphate buffer, pH 6.9 containing 0.02% CaCl2) 200 µL of yogurt extract and 400 µL buffer, incubated at 37°C for 10 min, then reacted with 200 µL 1 % soluble starch in buffer. After 10 min, 400 µL DNS (12 g dinitrosalicylic acid, 18.6 g NaOH, 305 g potassium sodium tartrate in 1L H2O) reagent was added, boiled for 5 min, cooled, and absorbance measured at 540 nm (BioMate 3S, Thermo Fisher Scientific, USA). Included blanks (no enzyme) and positive control (0.1-1 mM acarbose).
Sensory evaluation
Sensory evaluation was conducted by ten trained panelists, selected through visual and mental acuity screening and subsequently trained to assess variation in flavor, texture, color and overall appearance. The impact of adding MFDP to these sensory traits was examined using a 9-point hedonic scale.
Sensory stability
All the physicochemical analyses mentioned above were also conducted on fresh samples and after 10 and 15 days of storage at 4 ± 1°C under controlled conditions to evaluate the product’s shelf life.
Microbial viability
The viability of Lactobacillus bulgaricus and Streptococcus thermophilus in MFDP fortified yogurt was evaluated following Moura, et al. 2015. Samples were stored at two conditions: Room temperature (25 ± 2°C) and refrigeration (4 ± 1°C). Each sample was homogenized using a vortex mixer (Vortex Genie 2, Scientific industries, USA) set at 2000 rpm for 60 seconds. Serial dilution was done at (10-1-10-3) using 0.1% sterile peptone. Aliquots (0.1 ml) were spread on MRS agar for L. bulgaricus and M17 agar for S. thermophilus for 24 h aerobically. Colonies were counted, and results expressed as log cfu/ml. The colony counts were then converted to log cfu/ml using the Formula 4.

Microstructural analysis
The microstructure of MFDP-fortified yogurt was examined using SEM following Zhou et al. 2007. Samples (2-3 mm cubes) were fixed overnight in 4% glutaraldehyde (in 1.0 M phosphate buffer, pH 7.0), rinsed twice with the same buffer (5 min each), dehydrated through a graded ethanol series (50-100%, 20 min each), and subjected to drying. Samples were subjected to critical point drying, mounted on aluminum stubs, and sputter-coated with gold using an Emitech K850 (Quorum Technologies, UK). Imaging was performed using a Tescan VEGA LMU variable-pressure SEM (Tescan, Brno, Czech Republic) operated at 30 kV under high vacuum in SE mode. Images were captured at 3500 x magnification.
Statistical analysis
The study was assessed using an RCBD (Randomized Complete Block Design). Two-way ANOVA was employed to determine the significance level (p-value<0.05) of differences among the means of various factors. Statistical package for the social sciences version 23 and R programming language version 4.3.1 were utilized for statistical analysis to explore significant mean variances. Various types of graphs were employed to represent the data visually.
Results and Discussion
Physicochemical profile of MFDP fortified yogurt
Fortification of yogurt with MFDP resulted in pronounced and statistically significant changes in physicochemical characteristics across all the storage period as represented in Tab 1. The most evident changes were observed between the control (T0) and the highest fortification level (T4), indicating a clear concentration-dependent response. Total solids increased substantially with MFDP addition. At day 0, the control yogurt exhibited the lowest solids content (T0=13.5 %), whereas yogurt fortified with 2 % MFDP (T4) showed 22.57 %, However, a slight decrease was observed during storage. In contrast, moisture content ranged from 86.5 % in the control to 77.3 % in T4 at day 0 and showed a minor increase during storage across all treatments. Crude fiber was absent in the control yogurt but increased progressively with MFDP inclusion. Protein content showed a significant concentration-dependent increase in MFDP fortified yogurt treatments, whereas fat content remained statistically unchanged across all treatments and storage periods. Ash content increased significantly with increasing MFDP concentration, reflecting enhanced mineral incorporation into yogurt. Storage time significantly affected acidity-related parameters. A gradual decrease in pH and a corresponding increase in titratable acidity were observed in all treatments throughout storage. Syneresis increased during storage in all samples; however, MFDP fortified yogurts consistently exhibited lower whey separation compared to the control. Vitamin C content was markedly enhanced in MFDP fortified yogurts and remained relatively stable during storage. Overall, MFDP fortification, particularly at higher inclusion levels, significantly improved the nutritional composition, antioxidant-related attributes, and physical stability of MFDP fortified yogurt while maintaining acceptable physicochemical changes during storage.
| Treatments | MFDP fortified yogurt | |||
|---|---|---|---|---|
| 0 Days | 10 Days | 15 Days | ||
| Total solid content (%) | ||||
| T0 | 13.5 ± 0.1f | 13.3 ± 0.1fg | 13.02 ± 0.01g | |
| T1 | 17.8 ± 0.4e | 17.7 ± 0.1e | 17.43 ± 0.03e | |
| T2 | 18.77 ± 0.05d | 18.78 ± 0.05d | 18.61 ± 0.04d | |
| T3 | 21.01 ± 0.01b | 20.75 ± 0.05bc | 20.63 ± 0.03c | |
| T4 | 22.57 ± 0.15a | 22.5 ± 0.1a | 22.43 ± 0.01a | |
| Moisture content (%) | ||||
| T0 | 86.5 ± 0.19c | 86.96 ± 0.03b | 87.66 ± 0.01a | |
| T1 | 82.16 ± 0.05f | 82.63 ± 0.01e | 83.18 ± 0.04d | |
| T2 | 81.02 ± 0.09h | 81.22 ± 0.04h | 81.92 ± 0.03g | |
| T3 | 79.16 ± 0.05k | 79.58 ± 0.02j | 80.03 ± 0.04i | |
| T4 | 77.3 ± 0.1n | 77.83 ± 0.07m | 78.23 ± 0.03l | |
| Crude fiber (%) | ||||
| T0 | 0b | 0b | 0b | |
| T1 | 0.02 ± 0.01a | 0.02 ± 0.01a | 0.01 ± 0.01a | |
| T2 | 0.04 ± 0.01a | 0.04 ± 0.01a | 0.03 ± 0.01b | |
| T3 | 0.06 ± 0.01a | 0.06 ± 0.01a | 0.05 ± 0.01b | |
| T4 | 0.08 ± 0.01a | 0.08 ± 0.01a | 0.07 ± 0.01b | |
| pH | ||||
| T0 | 4.44 ± 0.01a | 4.19 ± 0.01b | 4.03 ± 0.01cd | |
| T1 | 4.26 ± 0.05b | 4.05 ± 0.04c | 3.87 ± 0.03ef | |
| T2 | 4.24 ± 0.03b | 4.03 ± 0.01cd | 3.85 ± 0.01ef | |
| T3 | 4.22 ± 0.01b | 3.99 ± 0.04cd | 3.81 ± 0.05fg | |
| T4 | 4.17 ± 0.01b | 3.94 ± 0.01de | 3.74 ± 0.01g | |
| Acidity (%) | ||||
| T0 | 0.87 ± 0.04j | 0.92 ± 0.01i | 1.01 ± 0.01h | |
| T1 | 1.08 ± 0.01g | 1.13 ± 0.02ef | 1.21 ± 0.02bc | |
| T2 | 1.10 ± 0.01fg | 1.15 ± 0.01de | 1.23 ± 0.01b | |
| T3 | 1.12 ± 0.01f | 1.17 ± 0.01d | 1.24 ± 0.01ab | |
| T4 | 1.13 ± 0.02ef | 1.18 ± 0.01cd | 1.26 ± 0.02a | |
| Ash content (%) | ||||
| T0 | 1.21 ± 0.01c | 1.19 ± 0.04c | 1.17 ± 0.02c | |
| T1 | 1.41 ± 0.03bc | 1.39 ± 0.01bc | 1.37 ± 0.04bc | |
| T2 | 1.71 ± 0.01ab | 1.69 ± 0.04ab | 1.67 ± 0.01ab | |
| T3 | 1.72 ± 0.02ab | 1.7 ± 0.05ab | 1.68 ± 0.05ab` | |
| T4 | 2.01 ± 0.01a | 1.99 ± 0.01a | 1.96 ± 0.01a | |
| Fat content (%) | ||||
| T0 | 3.51 ± 0.01a | 3.48 ± 0.02a | 3.50 ± 0.06a | |
| T1 | 3.48 ± 0.04a | 3.50 ± 0.04a | 3.47 ± 0.02a | |
| T2 | 3.51 ± 0.04a | 3.48 ± 0.01a | 3.48 ± 0.02a | |
| T3 | 3.47 ± 0.02a | 3.47 ± 0.04a | 3.50 ± 0.03a | |
| T4 | 3.47 ± 0.03a | 3.51 ± 0.01a | 3.47 ± 0.05a | |
| Protein Content (%) | ||||
| T0 | 5.81 ± 0.01h | 5.81 ± 0.03h | 5.80 ± 0.01h | |
| T1 | 6.21 ± 0.02g | 6.17 ± 0.01fg | 6.13 ± 0.03f | |
| T2 | 6.67 ± 0.03e | 6.66 ± 0.02e | 6.62 ± 0.02e | |
| T3 | 7.28 ± 0.01d | 7.25 ± 0.01cd | 7.23 ± 0.01bcd | |
| T4 | 7.37 ± 0.01a | 7.35 ± 0.05abc | 7.31 ± 0.01ab | |
| Syneresis (ml) | ||||
| T0 | 1.51 ± 0.01j | 1.87 ± 0.01f | 2.49 ± 0.01a | |
| T1 | 1.45 ± 0.04k | 1.85 ± 0.05fg | 2.38 ± 0.05b | |
| T2 | 1.39 ± 0.01l | 1.83 ± 0.03gh | 2.29 ± 0.03c | |
| T3 | 1.35 ± 0.05m | 1.80 ± 0.01hi | 2.25 ± 0.04d | |
| T4 | 1.30 ± 0.06n | 1.78 ± 0.03i | 2.20 ± 0.01e | |
| Vitamin C (mg AAE/100 g) | ||||
| T0 | 1.20 ± 0.02f | 1.19 ± 0.02g | 1.18 ± 0.01g | |
| T1 | 8.54 ± 0.03de | 8.54 ± 0.01de | 8.52 ± 0.01e | |
| T2 | 8.58 ± 0.02c | 8.57 ± 0.01c | 8.55 ± 0.03cd | |
| T3 | 8.62 ± 0.02b | 8.60 ± 0.04b | 8.57 ± 0.01c | |
| T4 | 8.65 ± 0.03a | 8.63 ± 0.01b | 8.60 ± 0.03b | |
| Note: T0 (Control, 0% MFDP), T1 (0.5% MFDP), T2 (1.0% MFDP), T3 (1.5% MFDP), T4 (2% MFDP). Values are mean ± SD; different superscripts denote significant differences as per Tukey’s test (p<0.05) | ||||
Table 1: Physicochemical properties of yogurt fortified with different concentration MFDP.
Color analysis of MFDP fortified yogurt
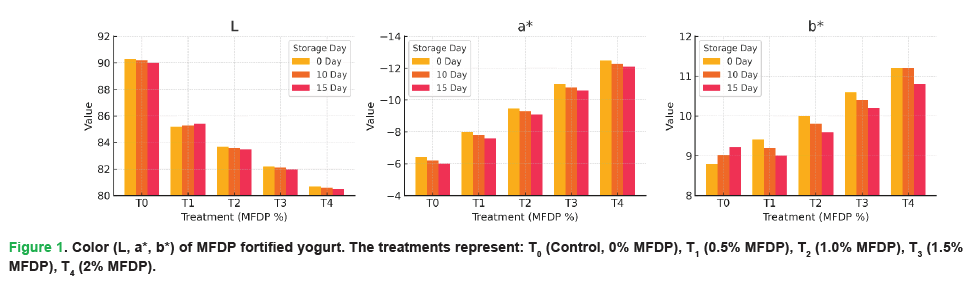
The color characteristics (L, a* and b*) of yogurt supplemented with varying levels of MFDP exhibited significant (p<0.05) alterations during storage, as shown in Fig 1. The lightness (L*) values showed a progressive decline from T0 to T4, indicating a darker appearance with higher MFDP incorporation, likely due to the natural green pigmentation of Moringa leaf. Concurrently, the a* values became more negative, reflecting enhanced greenness, while b* values increased, signifying greater yellowness, particularly at higher MFDP concentrations. These color modifications remained consistent throughout the storage period, with only minor variations over time.
Figure 1: Color (L, a*, b*) of MFDP fortified yogurt. The treatments represent: T0 (Control, 0% MFDP), T1 (0.5% MFDP), T2 (1.0% MFDP), T3 (1.5% MFDP), T4 (2% MFDP).
Mineral content of MFDP fortified yogurt
The mineral composition of MFDP fortified yogurt was evaluated over a storage period. The results demonstrated a clear and statistically significant enhancement (p<0.05) in mineral content with increasing MFDP inclusion levels, while storage duration exerted no significant effect on mineral stability. Calcium content increased progressively with MFDP fortification, ranging from 120 mg/100 g in the control (T0) to 200 mg/100 g in T4, with statistically distinct differences among treatments (p<0.05). Notably, calcium concentration remained stable throughout the 15-day storage period across all treatments. Similarly, phosphorous content exhibited a significant-treatment dependent increase, with values rising from 95 mg/100 g (T0) to 115 mg/100 g (T4). No significant variation was observed over storage time, indicating effective mineral retention within the yogurt matrix. A consistent and significant increase (p<0.05) in sodium content was observed as MFDP concentration increased, with values ranging from 45 mg/100g (T0) to 85 mg/100g in T4, while remaining stable over storage. Magnesium level followed a similar trend, increasing from 12 mg/100 g in the control to 44 mg/100 g in T4, with no statistically significant changes during storage. Iron content increased significantly (p<0.05) with MFDP incorporation, rising from 0.10 mg/100 g in T0 to 1.30 mg/100 g in T4. Although a slight numerical decrease was observed during storage, these changes were not statistically significant. Zinc concentration also exhibited a significant enhancement with fortification, increasing from 0.6 mg/100 g (T0) to 1.4 mg/100 g (T4), with consistent values maintained throughout the storage period. Overall, the results indicate that MFDP fortification substantially improves the mineral profile of yogurt in dose-dependent manner, without compromising mineral stability during refrigerated storage up to 15 days.
Antioxidant potential of MFDP fortified yogurt
The incorporation of MFDP markedly enhanced the antioxidant properties of yogurt. A significant (p<0.05) increase in total phenolic content and total flavonoid content was observed with increasing MFDP concentration across all storage intervals as shown in Fig. 2. At day 0, TPC increased from 12.61 ± 0.01 mg GAE/100g in the control (T0) to 28.13 ± 0.01 mg GAE/100g in the 2% MFDP treatment (T4). Similarly, TFC raised from 11.05 ± 0.05 mg RE/100g (T0) to 28.52 ± 0.02 mg RE/100g (T4). Although a gradual decline in both parameters occurred during storage, the fortified samples consistently maintained higher values than the control. Antioxidant potential, evaluated through DPPH, FRAP and TAC, also demonstrated a significant concentration-dependent increase with MFDP fortification. DPPH value increased from 20.81 ± 0.01% in the control to 75.32 ± 0.01% in T4 at day 0, indicating a strong free radical scavenging effect. Similarly, FRAP values increased from 39.50 ± 0.1 to 69.20 ± 0.1 µmol Fe+2/ 100g, while TAC improved from 60.4 ± 0.01 to 64.4 ± 0.02 mg AAE/100g over the same treatments. Although slight reductions were noted during storage, all fortified samples maintained significantly higher antioxidant activities compared to the control (p<0.05). These outcomes confirm the substantial contribution of MFDP to the enhancement of yogurt antioxidant profile, likely attributable to its rich polyphenolic and flavonoid composition. The retention of antioxidant capacity during storage further suggests good stability of MFDP-derived bioactive within the yogurt matrix compared to literature values. Overall, the results validate MFDP as an effective functional additive capable of improving the bioactive quality and oxidative stability of yogurt.
Figure 2: Effect of MFDP concentration on antioxidant profile of fortified yogurt. The treatments represent: T0 (control, 0% MFDP), T1 (0.5% MFDP), T2 (1.0% MFDP), T3 (1.5% MFDP), T4 (2% MFDP).
Alpha amylase activity of MFDP fortified yogurt
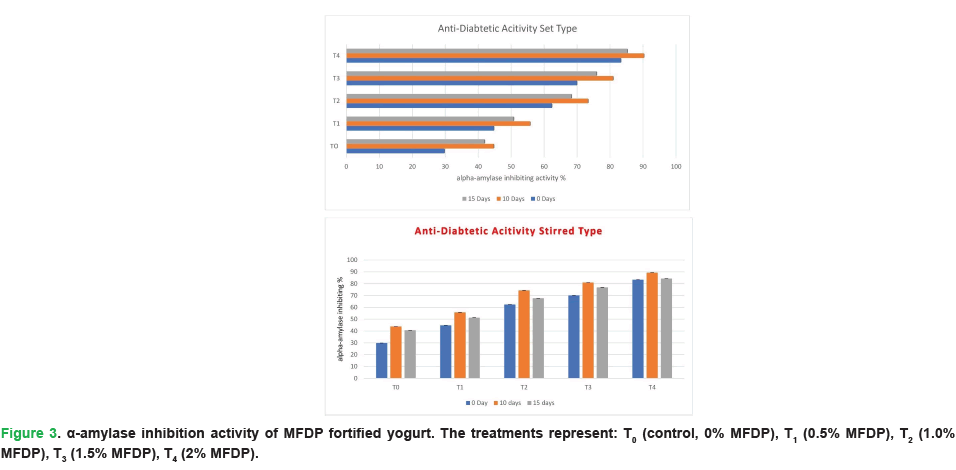
In recent years, there has been growing interest in the development of alternative therapeutic approaches for type-2 diabetes, particularly exploring phytochemicals that can impede or prevent glucose absorption. In the current study, the incorporation of MFDP into yogurt demonstrated a marked enhancement in alpha amylase inhibitory activity. The inhibition percentage increased with higher MFDP concentrations, reaching its peak on day 10 and declining slightly by day 15. Specifically, alpha amylase inhibition ranged from 29.80 ± 0.01% in the control (T0) and 85.32 ± 0.02 % in the highest fortified sample (T4) (Fig. 3).
Figure 3: α-amylase inhibition activity of MFDP fortified yogurt. The treatments represent: T0 (control, 0% MFDP), T1 (0.5% MFDP), T2 (1.0% MFDP), T3 (1.5% MFDP), T4 (2% MFDP).
The incorporation of MFDP into yogurt not only imparted functional health benefits but also contributed positively to product quality attributes.
Organoleptic evaluation of MFDP fortified yogurt
The organoleptic evaluation of MFDP fortified yogurt revealed significant (p<0.05) variations among treatments during 15 days of storage at 5°C. Color and flavor scores ranged from 7.31 ± 0.22 (T1) to 5.22± 0.29 (T4) as represented in Fig. 4, indicating a gradual decline in sensory appeal with higher MFDP concentrations. However, treatment T3 (1.5% MFDP) received the highest overall acceptability, attributed to its balanced flavor, texture and color stability.
Figure 4: Organoleptic evaluation of MFDP fortified Yogurt. The treatments represent: T0 (control, 0% MFDP), T1 (0.5% MFDP), T2 (1.0% MFDP), T3 (1.5% MFDP), T4 (2% MFDP).
Microbial viability of MFDP fortified yogurt
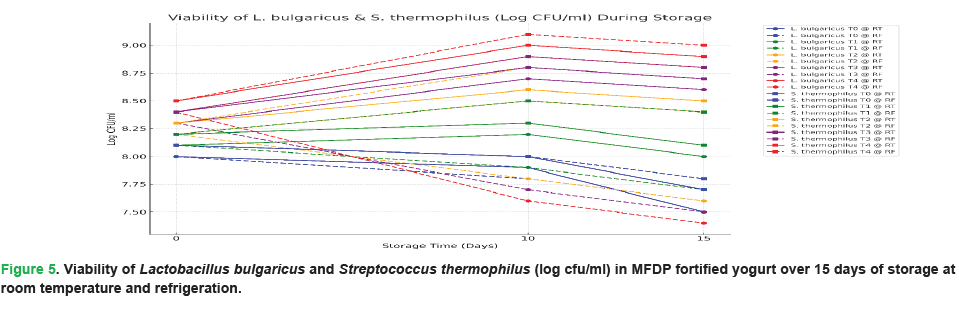
The viability of Lactobacillus bulgaricus and Streptococcus thermophilus in MFDP fortified yogurt was assessed over 15 days of storage period at room and refrigerated temperatures. As shown in Fig. 5, MFDP addition significantly enhanced probiotic survival compared to the control (T0), with the highest viability observed in T4 (2% MFDP). For L. bulgaricus, T4 exhibited a rise from 8.4 ± 0.02 to 8.9 ± 0.02 log cfu/ml by day 10, followed by a minor decline by day 15, whereas the control showed a steady decrease. A similar trend was noted for S. thermophilus, with T4 maintaining higher counts (9.0 ± 0.01 log cfu/ml) across all intervals. Refrigerated storage better preserved bacterial viability, through minor reductions occurred over time. The improved survival is attributed to the bioactive constituents of Moringa, particularly phenolics, amino acids, and peptides, which may act as prebiotic substrates and oxidative protectants, promoting microbial stability (Fig. 5).
Figure 5:Viability of Lactobacillus bulgaricus and Streptococcus thermophilus (log cfu/ml) in MFDP fortified yogurt over 15 days of storage at room temperature and refrigeration.
Microstructural analysis of MFDP fortified yogurt using SEM
Consumers generally prefer dense, high-quality yogurt with minimal serum separation, which is considered a defect that can be mitigated through structural optimization. To evaluate the influence of MFDP on yogurt microstructure, treatment T3 (1.5% MFDP) was compared with the control. Scanning electron microscopy at x3500 magnification (10-50 µm depth) revealed (Fig. 6) that the T3 yogurt exhibited a denser, more uniform matrix with reduced pore size and enhanced protein cross-linking compared to the control, which showed irregular and porous structures. ImageJ analysis confirmed smaller pore spaces (3.83 µm) and compact networks in the MFDP fortified sample, indicating improved gel integrity.
Figure 6: SEM image of MFDP Fortified Yogurt in 10 μm and 50 μm. Note: A and B: Control yogurt; C and D: MFDP fortified yogurt.
Correlation of physicochemical and bioactive components in MFDP fortified yogurt
The heat map (Fig. 7) represents the correlation matrix among various physicochemical, nutritional, and sensory parameters of MFDP fortified yogurt. The color gradient from blue to red indicates the strength and direction of correlations, where deep red denotes a strong positive correlation and deep blue indicates a strong negative correlation. The analysis reveals that antioxidant-relatedparameters (TPC, TFC, DPPH, FRAP, and TAC) exhibit strong positive correlations with minerals (iron, zinc, magnesium, phosphorous, and calcium), suggesting that MFDP fortification enhances both antioxidant activity and mineral enrichment. Moreover, overall acceptability, taste, flavor, and texture show positive association with color and moisture, indicating that these sensory attributes are significantly influenced by compositional quality. Conversely, pH and syneresis show negative correlations with antioxidant and mineral parameters, implying that higher MFDP concentration, while beneficial for bioactivity, may slightly lower pH and increase gel firmness, reducing whey separation. Cluster formation in the dendrogram suggests that antioxidant capacity and mineral enrichment group together, while sensory attributes and compositional factors from another cluster, demonstrating the dual influence of MFDP addition on both functional and sensory quality. Overall, the correlation structure supports the conclusion that MFDP fortification improves yogurt’s nutritional and antioxidant potential without severely compromising sensory acceptability, particularly at moderate inclusion levels.
Figure 7: The impact of MFDP on the physicochemical, nutritional, and antioxidant properties of yogurt treatments demonstrated through Heat Map.
Discussion
Fortification of yogurt with MFDP significantly improved its physicochemical, nutritional, antioxidant, and functional attributes, establishing MFDP as a next-generation natural fortificant with superior performance compared to conventional Moringa leaf powders or solvent extracts. The concentration-dependent increase in total solids, protein, and ash content across treatments reflected the nutrient richness of MFDP matrix, while the concurrent decrease in moisture and syneresis indicated enhanced gel stability fat levels and moderate acidity, demonstrating that MFDP fortification preserved the structural and sensory integrity of yogurt while substantially elevating its nutritional density. Unlike traditional Moringa leaf powder fortification, which often compromises textural uniformity and accelerates whey separation, the freeze-dried matrix facilitated improved protein-polyphenol cross-linking and microstructural compaction, leading to a denser and more cohesive gel network (El-gammal, et al. 2017). The stability of pH and the reduced rate of post-acidification during storage further highlights MFDP buffering potential, likely mediated by phenolic-protein interaction that modulate microbial acidification, resulting in a more stable product. The mineral composition of fortified yogurt exhibited a pronounced and stable enhancement, with calcium, phosphorus, magnesium, zinc and iron concentrations increasing markedly as MFDP inclusion rose (Ali, et al. 2019). These findings demonstrate that the MFDP not only delivers superior mineral density but also ensures retention during storage, likely due to the presence of organic chelators and amino acids that promote mineral complexation and prevent precipitation. Compared with prior studies using aqueous or ethanolic Moringa extracts, where nutrient losses and mineral instability were frequent, the MFDP matrix achieved consistent retention and bio accessibility, confirming its technological advantage as a natural and stable mineral fortificant (Aslam, et al. 2021).
The antioxidant analysis further substantiated MFDP’s superiority, revealing substantial increases in total phenolic content, total flavonoid content, and radical scavenging capacity (DPPH, FRAP, TAC). The freeze-drying process preserved the sensitive compounds such as quercitin, cholorogenic acid, and kaempferol, which are often degraded during solvent extraction or thermal drying, thereby maintaining antioxidant potency far beyond levels reported in Moring powder or extract-based yogurt. The high antioxidant retention validates MFDP’s ability to stabilize bioactive molecules and mitigate oxidative degradation, leading to improved product shelf life and functional efficacy. The strong positive correlation observed between antioxidant indices and mineral content underscores a synergistic enhancement of nutritional and bioactive potential. Additionally, the significant increase in alpha-amylase inhibitory activity highlights MFDP’s potential in developing functional dairy foods with anti-hyperglycemic properties. This effect can be attributed to stable polyphenolic compounds such as rutin and chlorogenic acid, which are known inhibitors of carbohydrate-hydrolyzing enzymes, and whose retention is maximized through freeze drying. Unlike ethanolic extracts, where enzyme inhibition declines sharply over time, MFDP maintained functional activity during storage, illustrating its advantage as a metabolically active, shelf-stable fortificant.
Microbial viability results further reinforced MFDP’s dual role as a nutrient source and bio-protectant. The enhanced survival of Lactobacillus bulgaricus and Streptococcus thermophilus suggests that MFDP provides bioavailable amino acids and phenolics, that supports microbial resilience, resulting in higher viable counts during storage. This prebiotic synergy enhances probiotic stability and healthpromoting potential, distinguishing MFDP-fortified yogurts from those fortified with raw leaf powders or extracts, where phenolic imbalance often inhibit bacterial growth (Shehata, et al. 2019). From a sensory and structural standpoint, the 1.5% MFDP formulation achieved the optimal balance between bifunctional enhancement and consumer acceptability, maintaining desirable color, texture, and flavor attributes despite the natural green pigmentation. Scanning electron microstructure, revealing smaller pore sizes and compact protein networks consistent with reduced syneresis and improved gel strength. Collectively, these findings establish MFDP as a technologically advanced and functionally superior fortificant, capable of improving the compositional quality, oxidative stability, and probiotic performance of yogurt while maintaining sensory acceptance. In contrast to conventional Moringa powders or extract, which often face limitations in stability, solubility, and flavor, the freeze-dried extract powder preserves phytochemical complexity, nutrient integrity, and bio-functional efficacy, offering a holistic advantage for functional dairy development. This study therefore provides the first comprehensive evidence that MFDP not only enhances nutritional and antioxidant capacity but also ensures longterm biochemical and microbial stability, positioning it as a novel, clean-label ingredient for next-generation functional foods aimed at health optimization and metabolic wellness.
Conclusion
The incorporation of MFDP significantly enhanced the physicochemical, antioxidant, and functional attributes of yogurt. Fortification with MFDP not only improved bioactive potential, reflected by elevated phenolic and flavonoid content, enhanced antioxidant capacity, and greater alpha-amylase inhibition, supporting its role as a natural antidiabetic ingredient. The addition of MFDP also fortifies yogurt with essential minerals and improves its microstructural integrity, color attributes, and sensory acceptability, particularly at 1.5% (T3) concentration. Importantly, the viability of Lactobacillus bulgaricus and Streptococcus thermophilus remained stable, confirming that MFDP integration does not compromise fermentation performance. These outcomes collectively highlight MFDP as a multifunctional ingredient capable of transforming conventional yogurt into a health-promoting functional food with potential benefits for glycemic regulation and metabolic wellness. Future studies should explore the bioavailability of Moringaderived compounds and their synergistic interaction within complex food matrices to broaden industrial applications.
Acknowledgment
We acknowledge Dr. Amna Saeed, for facilitation regarding proofreading assistance for the article.
Funding
No organization funded this study.
Conflict of Interest
The authors declare no conflict of interest regarding this manuscript.
References
- Adepoju FO, Selezneva IS. (2024). Production and evaluation of set-type yoghurt fortified with chlorella vulgaris and Moringa oleifera powder. Plant Foods Hum Nutr. 79:531-538.
[Crossref] [Google Scholar] [PubMed]
- Ali A, Yusof YA, Chin NL, Ibrahim MN, Basra SMA. (2019). Proximate composition of Moringa oleifera products from Pakistan. Pak J Bot. 51:1025-1032.
- AOAC international. (2019). Official methods of analysis (21st ed.).
- Aslam M, Anwar F, Nadeem R, Rashid U, Kazi TG, Nadeem M. (2005). Mineral composition of Moringa oleifera leaves and pods from different regions of Punjab, Pakistan. Asian J Plant Sci. 4:417-421.
- Bankole PO, Omoni VT, Tennison-Omovoh CA, Adebajo SO, Mulla SI. (2023). Enhanced removal of dibutyl phthalate in a laccase-mediator system: Optimized process parameters, kinetics, and environmental impact. J Environ Manage. 348:119227.
[Crossref] [Google Scholar] [PubMed]
- Bhatta R, Pandey S, Gurung S, Hossain A. (2025). Effect of moringa leaf meal supplementation on growth performance and survival of mono sex tilapia (Oreochromis Niloticus) in cage culture during winter season. Int J Fish Aquac. 17:39-47.
- DiniÄ M, JakovljeviÄ S, PopoviÄ N, RadojeviÄ D, VeljoviÄ K, GoliÄ N, TerziÄ-VidojeviÄ A. (2023). Assessment of stability and bioactive compounds in yogurt containing novel natural starter cultures with the ability to promote longevity in Caenorhabditis elegans. J Dairy Sci. 106:7447-60.
[Crossref] [Google Scholar] [PubMed]
- El-Gammal RE, Abdel-Aziz ME, Darwish MS. (2017). Utilization of aqueous extract of Moringa oleifera for production of functional yogurt. J Food Dairy Sci. 8:45-53.
- Fatima HN, Maryam H, Jamil MU. (2026). Development and characterization of chitosan-amoxicillin hydrogel for treating MRSA-infected burn wounds in a murine model. Bull Biol All Sci Res. 2026:113.
- Fitriana AS, Royani S. (2020). Identifying antioxidant activities of guava fruit using DPPH method. In1st International Conference on Community Health (ICCH 2019). Atlantis Press. 113-115.
- Freitas M, Proença C, Ribeiro D, Quinaz-Garcia MB, Araújo AN, Fernandes E. (2023). Assessment of α-amylase activity in a microanalysis system: Experimental optimization and evaluation of type of inhibition. J Chem Educ. 100:1237-1245.
- Ghafar F, Nazrin TT, Salleh M, Hadi NN, Ahmad N, Hamzah AA, Yusof ZM, Azman IN. (2017). Total phenolic content and total flavonoid content in Moringa oleifera seed. Galeri Waris Sains. 1:23-35.
- Granato D, Barba FJ, BursaÄ KovaÄeviÄ D, Lorenzo JM, Cruz AG, Putnik P. (2020). Functional foods: Product development, technological trends, efficacy testing, and safety. Annu Rev Food Sci Technol. 11:93-118.
[Crossref] [Google Scholar] [PubMed]
- Hashemi M, Hashemi M, Amiri E, Hassanzadazar H, Daneshamooz S, Aminzare M. (2019). Evaluation of the synergistic antioxidant effect of resveratrol and Zataria multiflora boiss essential oil in sodium alginate bioactive films. Curr Pharm Biotechnol. 20:1064-1071.
[Crossref] [Google Scholar] [PubMed]
- International Diabetes Federation (IDF). (2021). The diabetes atlas. IDF diabetes atlas (10th ed.). International Diabetes Federation.
- Kim MA, Kim MJ. (2020). Isoflavone profiles and antioxidant properties in different parts of soybean sprout. J Food Sci. 85:689-695.
[Crossref] [Google Scholar] [PubMed]
- Kim YH, Khan AL, Waqas M, Lee IJ. (2017). Silicon regulates antioxidant activities of crop plants under abiotic-induced oxidative stress: A review. Front Plant Sci. 8:256717.
[Crossref] [Google Scholar] [PubMed]
- Li X, Yadav R, Siddique KH. (2020). Neglected and underutilized crop species: The key to improving dietary diversity and fighting hunger and malnutrition in Asia and the Pacific. Front Nutr. 7:593711.
[Crossref] [Google Scholar] [PubMed]
- Lu Q, Jin L, Wang P, Liu F, Huang B, Wen M, Wu S. (2023). Effects of interaction of protein hydrolysate and arbuscular mycorrhizal fungi effects on citrus growth and expressions of stress-responsive genes (Aquaporins and SOSs) under salt stress. J Fungi (Basel). 9:983.
[Crossref] [Google Scholar] [PubMed]
- Luqman S, Srivastava S, Kumar R, Maurya AK, Chanda D. (2012). Experimental assessment of Moringa oleifera leaf and fruit for its antistress, antioxidant, and scavenging potential using in vitro and in vivo assays. Evid Based Complement Alternat Med. 2012:519084.
- Mehwish HM, Riaz Rajoka MS, Xiong Y, Zheng K, Xiao H, Anjin T, Liu Z, Zhu Q, He Z. (2022). Moringa oleifera-a functional food and its potential immunomodulatory effects. Food Rev Int. 38:1533-1552.
- Moura MC, Napoleão TH, Coriolano MC, Paiva PM, Figueiredo RC, Coelho LC. (2015). Waterâsoluble Moringa oleifera lectin interferes with growth, survival and cell permeability of corrosive and pathogenic bacteria. J Appl Microbiol. 119:666-676.
[Crossref] [Google Scholar] [PubMed]
- Mousavi S, Bereswill S, Heimesaat MM. (2019). Immunomodulatory and antimicrobial effects of vitamin C. Eur J Microbiol Immunol (Bp). 9:73-79.
[Crossref] [Google Scholar] [PubMed]
- Safdar R, Imran M, Mushtaq E, Nasir A. (2026). From toxicology to technology: Human health risks of mycotoxins in the food chain and current approaches to their detection and control. Bull Biol All Sci Res. 2026:114.
- Sankhyan NI, Sharma AN, Seth CA, Chauhan AN, Kulshrestha S. (2013). Determination and comparison of vitamin C content from Moringa oleifera by different methods. Int J Agric Sci Res. 3:67-70.
- Shehata MG, Abd-Rabou HS, El-Sohaimy SA. (2019). Plant extracts in probiotic encapsulation: Evaluation of their effects on strains survivability in juice and drinkable yogurt during storage and an in-vitro gastrointestinal model. J Pure Appl Microbiol. 13:609-617.
- Sonu K. (2023). Development of Moringa oleifera enriched flavoured milk and yoghurt and evaluation of physicochemical and bio-functional properties (Doctoral dissertation). National Dairy Research Institute.
- Stohs SJ, Hartman MJ. (2015). Review of the safety and efficacy of Moringa oleifera. Phytother Res. 29:796-804.
[Crossref] [Google Scholar] [PubMed]
- Vijay L, Karthikeyan N, Kumaresan G, Kathirvelan C. (2022). Sensory properties of functional Greek yoghurt incorporated with Moringa oleifera leaf powder. Pharma Innov J. 11:251-254.
- Zhou T, Yu H, Wang S. (2016). Effect of microstructural types on toughness and microstructural optimization of ultra-heavy steel plate: EBSD analysis and microscopic fracture mechanism. Mater Sci Eng A. 658:150-158.