Research - Modern Phytomorphology ( 2023) Volume 17, Issue 5
O. Fishchuk, Lesia Ukrainka Volyn National University, Pr. Voli. 13, Lutsk, 43025, Ukraine, Email: Dracaenaok@ukr.net
Received: 21-Jan-2021 Accepted: 27-Jan-2021 Published: 05-Mar-2021, DOI: 10.5281/zenodo.5801087
Abstract
We studied the structure of the flowers of the Cyrtanthus elatus (Jacq.) Traub. The morphometric parameters, morphology, anatomy, and vascular anatomy of the ovary and the flower are generally described. There are three vertical zones present in the C. elatus gynoecium: symplicate, hemisymplicate, and asymplicate vertical zones. Synascidiate zone is absent. Septal nectaries appear in the symplicate zone and have a height of about 1920 μm. C. elatus peduncle contains 20 vascular bundles, which unite above and form a massive vascular cylinder, from which at the level of the receptacle depart traces of dorsal vascular bundles, traces of septal vascular bundles, traces of tepals, which give rise to traces of stamens. In the center of the ovary remains a group of small vascular bundles-the roots of the ventral complex, which above when the locule appear reorganized into 6 massive vascular bundles-the ventral vascular bundles of the carpel, which feed the ovules. The trace of the ovule is single-bundle. Traces of dorsal and septal vascular bundles of the carpel are two-bundle. Above the locules, the ventral vascular bundles of the carpel are merge with the dorsal vascular bundles and form a dorsal vein. There are a lot of ovules, in a section in each locule are 2-3 ovules. Traces of the inner tepals comprise 10 vascular bundles, traces of the outer tepals comprise 15 vascular bundles. Traces of stamens are single-bundle, depart from tepal traces.
arion-krmiva kupispredas rottoconsultants hattrennet insurancemarketingpros abcoelectricli paddriver bobcoironrailings cliniquemtarhemodialyse gtech settechny groupe-saturne caffeitaliany rcollision husoghytteplan okba-medicaments mazex atyourserviceoil hetgroenewerk msgfeather rolltech ismllw lorlin conceriacaponigiuseppe chouikha-big dona-hotel ibrax fullthrottleeventplanning theblindspotli mustakynnys curdent auprintemps koulouritispolis floorbufferbrush minicoindustries thespongecompany localvisits fixcars baofoto poi elvisnewman palestragymtonic cavalierifuel unityrubberproductsllc menuiseriemorlighem nugris skillslab mecanica vksim brcanvas handfordoil lemi-yhdistys schoolbuspartsnow thebestofchampaign testingmechanics comm-unique goodnewsbooks generaldecor campye defence-institute fairwaymanorllc whpdc regionalsigns babuin islandeastdentalgroup sirajeslov adda thebestofcleveland integraff sealfiberglass nutechsys tcilandscaping digitaltechsquad tomsvetteshop funda-mantels rahaanopeasti thebowmanfirm marshallcoffeeny mrbrushes sicop-pentacol myguyappliance ldclean ephesusmedcuisine alphafastenersusa unitysurfacing allseasons-mechanical easyketodietsuccess liusaari strongarmcleaningny josg sterlingcutter unadesinfection huizebuitenhuis thebestofraleigh drcentralbaking catsol traditionaltrainsandhobbies goelectricnj casaconcreteinc thebestroofingcompanies autoproautomotiveservice general-machine djsensationalsounds fevaagbaatforening straightlinecustomconstruction impianti-antizanzare thebestoffortworth rosanneebner edandson americanchoirgown violiner jeromeandleighcrutch promisinguk iasautomotive scrittori plasticsrecycling giuseppedainelli jerryspride prontointerventoelettricista24h fimad gdezinewraps kimmokakko tracony fiavet bwnivelles alsalamzorg septimus pdcfundraising hoteldellaspina francobianchi cleansweepcremations errachid thebestofeugene grossoregistratori brotherspastries bellmoreglassandmirror plasmapreen microdecisionsystems eternoholdings alessiocostruzioni hotelpisa excelcourtreporters gmstow tatsrl chateaulamercatering bargaouirideau shcarwash shipritebags allportstrucking juventuspizza red-agri licorneargent thebestofphiladelphia lewisy greenrecuperi thebestofindianapolis fosenlagetsangkor coastweldingsupply nyfixcars alshubcaps hanssenspronkfamilierecht scottsafe barbatonursery jelconst jerryshulmanproduce orchardrealty minervasbandb dialindustries phytosif icredit thebestoftallahassee malermester-blakstad morvayautosiskola tektronicsinc cance-tu-asbl bicotec martinsqualitytruckbody paprikalongbridge roger-jensen rockypointbarbershop rmkdistributors tommiriiulid atlaschemicalllc gopaverinstaller onyxchb thewindowmill khalfallah-pneus ciaociao lexilogistics tipografiaelleemme thebestofwichita cdvdpro roll-n-roaster repelrestoration kcfapi ivar-moe bacosport byggkonsult predicate rohanengineeringpc footpharmacydirect solidbox piovesan visserijverduurzaamt toscanibus sixgsroofing tecnocostruzionizella outsourcemarketingpros pilotexamsdgca carraihome royalbakersdist guardiedicitta bestbaby-tn littlechicken werks1inc hodsonoilco surfacingsystems lckcabinetry pbtools4u set-mfg serristoricountry goldenmoonusa fourcmanagement lioutdoorliving nova-euro-fashion lavecchiacascina pomaraf novamaille dkstechnoholdings liisasauso sahel-tunisie inspired-tech aldamartini monarchengraving craldipendentiuslprato centuryhardware scholengroep ourtowncarwashandquicklube mayablog geometraparisi ash-grove tendertoo smithoilcompany westfrieslanddakbedekkingen lamaisonbeb blog ebiketime laurenty puurklant vincentwielders chams nassausuffolkirrigation bbdps aerocbt fursbysuperior unityrubbercompany horizonconceptinc immstema vhujon nativelandsmokeshop kaabia-orthodontie locali brooklynterminalmarketonline transportopplaering neonmazesl kuturanta apsbox federalnetworks liontrading power-tran degryse-chauffage uniquemasonrycorp proramps catchasilverstar projectbinder unityllc italyvacationpackages rrappliances norwestac topspintennisli boatfindertransport sblcollege imperialvendinginc nanea igiardinidibeatrice pslniemela holycowindian sisekosmosejaam acquadirete obertaberhof thebestofdallas betterheader mrpickleinc medspareparts thebestofsacramento toyotavandergeest alliance-consulting scbox sshoreendo thebestofgreenbay fiducia-partner komunikujeme lapetitemaisonenfrance chesterplastic wisesystems terveysverkko oportal cjflagandson gold-estate spiriolaw salvatorechiarelli autoskola trendcreditcorp scooterkingalmere sisternibedita thebestofminneapolis cerealism transnationalusa homedelano francobenvenutiartista kulsaasvelforening florencevillavioletta gettingerfeathers touchofclasscoll tomkovci
Keywords
Cyrtanthus elatus, ovary, gynoecium morphology, vertical zones, vascular bundles
Introduction
Genus Family Amaryllidaceae s.l. represented by a large number of ornamental plants, with large and bright flowers. Most members of this family grow in tropical and subtropical areas. The genus belongs to the family Amaryllidaceae J.St.-Hil., Subfamily Amaryllidoideae, tribe Cyrtantheae and has about 56 species (Chase et al. 2016). Details of the history, taxonomy, phylogeny, life cycle and cultivation features of Cyrtanthus taitii have been studied (Duncan 2018). New Zealand scientists tested for self-sterility in three Cyrtanthus species: C. contractus, C. ventricosus and C. mackenii by means of controlled hand‐pollination experiments. To determine the growth rates and frequency of ovule penetration using self-versus cross-pollen tubes, they used fluorescence microscopy to examine flowers collected at 24 h, 48 h and 72 h after pollination (Johnson et al. 2019). Pollination and late-acting self-incompatibility and implications for seed production in Cyrtanthus breviflorus (Amaryllidaceae) were studied (Vaughton et al. 2010). Development of an electrochemical biosensor for phylogenetic analysis of Amaryllidaceae based on the enhanced electrochemical fingerprint recorded from plant tissue was was investigated by P. Zhang and a group of researchers (Zhang et al. 2020). The aim of the work is to study the morphological features of the flower, gynoecium and septal nectaries of C. elatus, using micromorphology data in the taxonomy of the genera in Amaryllidaceae s.l.
Materials and Methods
Plant material was collected in the A.V. Fomin Botanical garden of Taras Shevchenko National University of Kyiv and fixed in 70% alcohol. For light microscope observations material was sectioned using standard methods of Paraplast embedding and serial sectioning at 20 mkm thickness (Barykina et al. 2004). Sections were stained with Safranin and Astra Blau and mounted in Eukitt. Digital photomicrographs were made using a microscope AMSCOPE T490B-10M (USA) and microscope digital camera AMSCOPE 10MP MU1000 (USA). Height of the gynoecium vertical zones was obtained after calculation of the transverse sections of the pistil.
We used the concept of vertical zonation of gynoecium by W. Leinfellner (Leinfellner 1950) to analyze the internal structure of the gynoecium, which takes into consideration only the congenital fusion of the carpels. According to this concept, with growth of the carpels, in the syncarpous gynoecium, congenital multilocular synascidiate, unilocular simplicate, transitional hemisimplicate and asymplicate (apocarpous) zones form. The methodological basis of the vertical zonation gynoecium study was formulated by W. Leinfellner, later the method was elaborated for monocots (Odintsova 2013).
Results
General flower morphology and micromorphology
C. elatus flowers up to 9 cm-12 cm long, slightly zygomorphic bright red. There are 5 flowers in the inflorescence. The flowers in C. elatus have a long flower tube, which is continued by six free tepals.
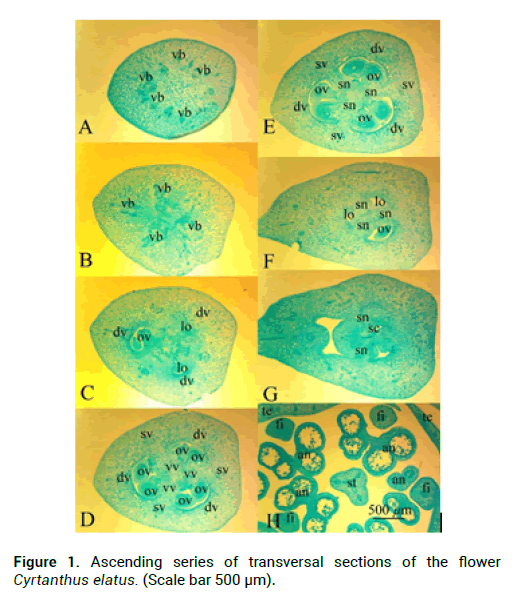
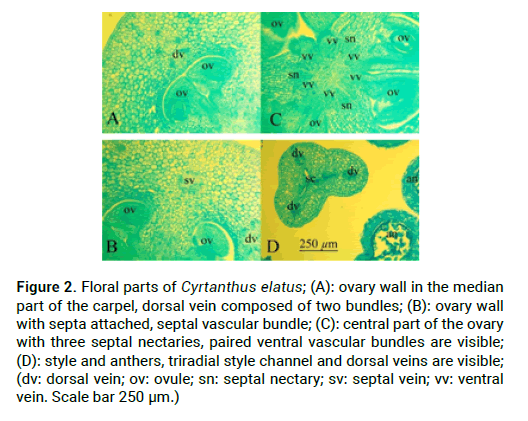
C. elatus stamens attached to the flower tube. Stamen filaments at the base are dilated, tapering to the apex, outer stamens are 7.1 cm-7.2 cm long and 0.1 cm in diameter, and inner stamens are 7 cm and 0.1 cm in diameter. Anthers are linear, introse, dorsifixed, anthers of external stamens are 0.5 cm long, anthers of internal stamens are 0.4 cm long (Fig. 1H). Ovary in C. elatus is inferior, green, oval, angular 1.3 cm long and 0.4 cm in diameter, which turns into a slightly zygomorphic, white at the base and red in the top, s-shaped style (Fig. 2D). In C. elatus gynoecium there are: a symplicate structural zone–700 μm (Fig. 1C and Fig. 1D), a hemisymplicate zone–1840 μm (Fig.1 and Fig. E). Asymplicate zone is absent. Septal nectary appears in the symplicate zone and continues in the hemisymplicate zone its total height is 2140 μm (Fig. 2C).
Figure 1: Ascending series of transversal sections of the flower Cyrtanthus elatus. (Scale bar 500 μm).
Figure 2: Floral parts of Cyrtanthus elatus; (Ð): ovary wall in the median part of the carpel, dorsal vein composed of two bundles; (B): ovary wall with septa attached, septal vascular bundle; (C): central part of the ovary with three septal nectaries, paired ventral vascular bundles are visible; (D): style and anthers, triradial style channel and dorsal veins are visible; (dv: dorsal vein; ov: ovule; sn: septal nectary; sv: septal vein; vv: ventral vein. Scale bar 250 μm.)
Flower anatomy
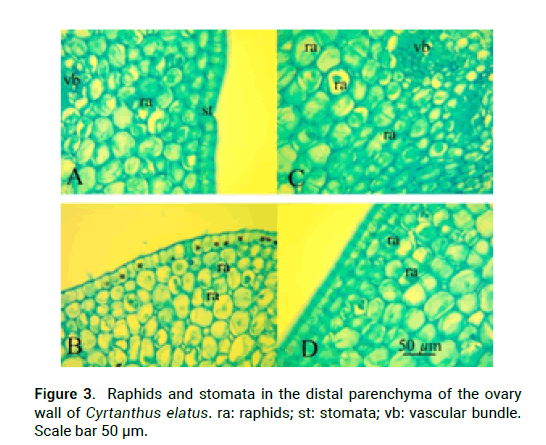
In the upper part of the peduncle, at the base of the flower tube, in the stamens and in the ovary wall there are idioblasts with cellular inclusions–raffids (Fig.3). They are absent in the free tepals, in connective and in the style. C. elatus peduncle contains 20 vascular bundles (Fig. 1A), which unite above and form a massive vascular cylinder, from which at the level of the receptacle depart traces of dorsal vascular bundles, traces of septal vascular bundles, traces of tepals, which give rise to traces of stamens (Fig. 1B). In the center of the ovary remains a group of small vascular bundles-the roots of the ventral complex, which above when the locule appear reorganized into 6 massive vascular bundles-the ventral vascular bundles of the carpel, which feed the ovules (Fig. 1C-E). The trace of the ovule is single-bundle (Fig. 1D). Traces of dorsal and septal vascular bundles (Fig. 2B) of the carpel are twobundle. Above the locules, the ventral vascular bundles of the carpel are merging with the dorsal vascular bundles and form a dorsal vein (Fig. 1H-G). There are a lot of ovules in a section in each locule are 2-3 ovules (Fig. 2A). Inner tepals include 10 vascular traces; outer tepals include 15 vascular traces. Traces of stamens are single-bundle; depart from tepal traces (Fig. 1H).
Figure 3: Raphids and stomata in the distal parenchyma of the ovary wall of Cyrtanthus elatus. ra: raphids; st: stomata; vb: vascular bundle. Scale bar 50 μm.
Discussion
Flowers in genus Cyrtanthus are actinomorphic to weakly zygomorphic, flower tube is usually longer than perianth segments. Filaments are biseriate, rarely united into a short corona. Stigma is deeply trifid to capitate (Meerow et al. 1998). In C. elatus gynoecium there are two structural zones: a symplicate and a hemisymplicate. C. elatus characterized by the presence 6 massive vascular bundles–the ventral vascular bundles of the carpel, which feed the ovules. The vascular system of the inferior ovary before was described in Galanthus nivalis L. and Leucojum vernum L. (Fishchuk and Odintsova 2020).
Conclusion
The study of the vascular anatomy of C. elatus and other members of the Amaryllidaceae family allows us to analyze the morphological features which laid in the fruit stage and confirm or refute the molecular distribution of genera in the Amaryllidaceae family, as there is a debate among scientists about the number of genera in subfamilies. The height of the septal nectary and the height of the different vertical ovary zones, gynoecium features and the correlation of all three types of vertical zonality allow us to more accurately differentiate the studied species and their morphological and vascular anatomy features that are unique.
References
Fishchuk O.S. (2020). Micromorphology and anatomy of the flower of galanthus nivalis and leucojum vernum (AMARYLLIDACEAE J. ST.-HIL.). Scientific Issue Ternopil Volodymyr Hnatiuk National Pedagogical University. Series: Bio 80: 26-33. http://journals.chem-bio.com.ua/index.php/biology/article/view/93
Chase M.W., Christenhusz M.J.M., Fay M.F., Byng J.W., Judd W.S., Soltis D.E., Stevens, P.F. (2016). An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Bot J Linnean Soc 181: 1-20. https://doi.org/10.1111/boj.12385
Duncan G. (2018). 870. Cyrtanthus Taitii: Amaryllidaceae. Curtis's Botanical Magazine 35: 24-35. https://doi.org/10.1111/curt.12222
Fishchuk O.S., Odintsova A.V. (2020). Micromorphology and anatomy of the flowers of Galanthus nivalis and Leucojum vernum (Amaryllidaceae). Regulat Mech Biosyst 11: 463-468. https://doi.org/10.15421/022071
Johnson S.D., Butler H.C., Robertson A.W. (2019). Breeding systems in Cyrtanthus (Amaryllidaceae): variation in self-sterility and potential for ovule discounting. Plant Biol 21: 1008-1015. https://doi.org/10.1111/plb.13025
Leinfellner W. (1950). The construction plan of the Syncarpian gynoceum. Aust Bot J 97: 403-436. https://www.jstor.org/stable/43336633
Meerow A.W., Snijman D.A. (1998). Amaryllidaceae. In K. Kubitzki [ed.], Families and genera of vascular plants. Springer 83-110.https://doi.org/10.17660/ActaHortic.2008.792.46
Odintsova A. (2013). Dva osnovnyx typy septalnyx nektarnykiv odnodolnyx. Visnyk Lvivskogo universytetu. Seriya biologichna 61: 41-50. https://doi.org/10.14202/vetworld.2016.231-234
Fu L., Zheng Y., Zhang P., Zhang H., Xu Y., Zhou J., Lin, C.T. (2020). Development of an electrochemical biosensor for phylogenetic analysis of Amaryllidaceae based on the enhanced electrochemical fingerprint recorded from plant tissue. Biosens Bioelect 159: 112212. https://doi.org/10.1016/j.bios.2020.112212